technology
What is the molecular geometry of nh2?
Written by Robert Harper — 0 Views
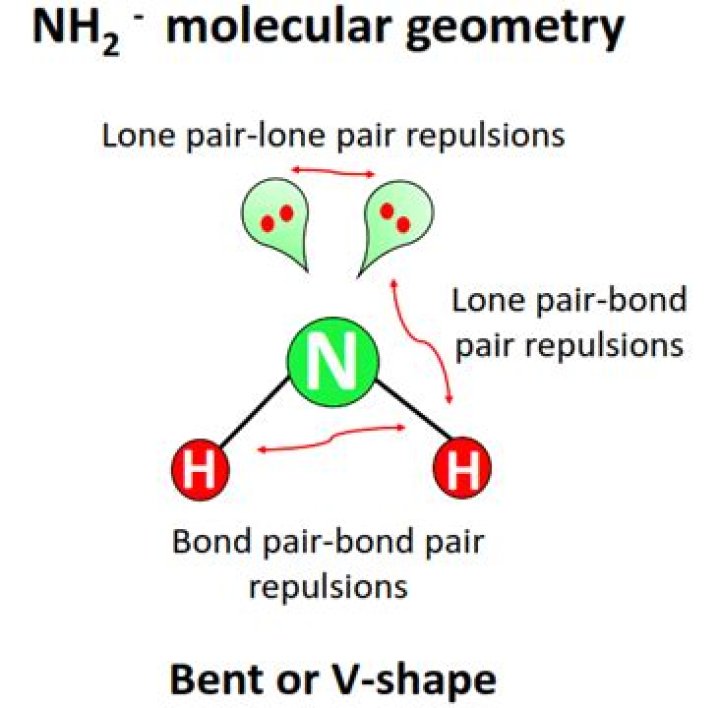
The overall shape would be bent (aka angular) with 104.5 degree bond angles due to strong lone pair-bonding pair repulsion that pushes the 2 bonding pairs closer together than they would like.
.
Also know, what shape is nh2?
the shape of NH2 is v-shaped or Bent shaped..
Additionally, what is the bond angle of nh2? science. NH2-, NH3, and NH4+ have H-N-H bond angles of 105, 107 and 109.
Also know, what is the Lewis structure of nh2?
Transcript: This is the NH2- Lewis structure. We have five valence electrons for nitrogen. Plus one for hydrogen, but we have two hydrogens then we need to add this extra valence electron in for a total of eight valence electrons for the NH2- Lewis structure.
What is the molecular geometry of co3 2?
We see from Figure 10.3. 3 that the molecular geometry of CO 3 2− is trigonal planar with bond angles of 120°.
Related Question AnswersWhat is the shape of c2h4?
trigonal planarWhat are the 5 basic shapes of molecules?
Molecular Geometries. The VSEPR theory describes five main shapes of simple molecules: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral.Is nh2 positive or negative?
The NH2 ion is NH2- - it has an extra electron to give it a -1 charge. So you are getting five electrons from the nitrogen, 1 from each hydrogen, and 1 extra to give a negative charge - total of eight electrons (octet around nitrogen).How many lone pairs are in ch4?
If these are all bond pairs the molecular geometry is tetrahedral (e.g. CH4). If there is one lone pair of electrons and three bond pairs the resulting molecular geometry is trigonal pyramidal (e.g. NH3). If there are two bond pairs and two lone pairs of electrons the molecular geometry is angular or bent (e.g. H2O).What is the shape of OCl2?
a. OCl2: V-shaped, polar; OCl2 is polar because the two O-Cl bond dipoles don't cancel each other.What is nh2 called in chemistry?
Azanide (chemical formula NH − 2) Amino radical (chemical formula NH •Is nh2 an acid or base?
The conjugate acid of NH2- is NH3, called ammonia. Ammonia is actually itself a weak base, so its conjugate base NH2- is an incredibly strong base so it can get an extra proton to regenerate NH3 which is much more stable.Is bf3 trigonal planar?
Molecular Geometry of BF3 The geometry of molecule of BF3 is 'Trigonal Planar. ' With the reference of Chemistry, 'Trigonal Planar' is a model with three atoms around one atom in the middle.What is the Lewis structure for c2h4?
Drawing the Lewis Structure for C2H For C2H4 you have a total of 12 total valence electrons. Drawing the Lewis structure for C2H4 (named ethene) requires the use of a double bond. In a double bond two pairs of valence electrons are shared (for a total of four valence electrons).How do you determine hybridization?
A Shortcut For Determining The Hybridization Of An Atom In A Molecule- Look at the atom.
- Count the number of atoms connected to it (atoms – not bonds!)
- Count the number of lone pairs attached to it.
- Add these two numbers together.
How do you find the formal charge?
Formal Charge = [# valence electrons on neutral atom] – [(# lone electron pairs) + (½ # bonding electrons)] Valence electrons = corresponds to the group number of the periodic table (for representative elements). Lone Pairs = lone electrons sitting on the atom. Each electron counts as one and so a pair counts as two.What is the molecular geometry of h2s?
Answer and Explanation: The molecular shape of hydrogen sulfide is bent. The central atom sulfur is bonded to two hydrogen atoms.What is the geometry of h2o?
tetrahedralWhat are the approximate HNH bond angles in nh4+?
The bond angles in ammonium ion, NH4^+ are the theoretical tetrahedral bond angles of approximately 109.5 degrees. The ammonium ion is completely symmetrical. The bond angles in the asymmetrical ammonia molecule are less than 109.5 degrees, they are about 107 degrees.What is the molecular geometry of CCl4?
tetrahedralIs co2 polar or nonpolar?
Carbon dioxide is non-polar because of the symmetry of its bonding. The electronegativity difference between carbon and oxygen is 1.0, which makes the bonds polar. However, the two polar bonds are at 180 degrees to each other so the dipoles cancel out.How do you find the molecular geometry?
Steps Used to Find the Shape of the Molecule- Draw the Lewis Structure.
- Count the number of electron groups and identify them as bond pairs of electron groups or lone pairs of electrons.
- Name the electron-group geometry.
- Looking at the positions of other atomic nuclei around the central determine the molecular geometry.



