What is esterification reaction give an example?
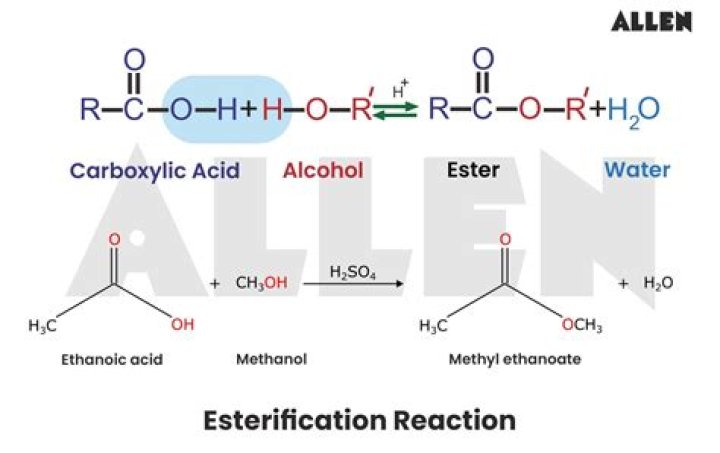
.
Keeping this in view, what is esterification give an example?
Some esters can be prepared by esterification, a reaction in which a carboxylic acid and an alcohol, heated in the presence of a mineral acid catalyst, form an ester and water: The reaction is reversible. As a specific example of an esterification reaction, butyl acetate can be made from acetic acid and 1-butanol.
Likewise, what is esterification reaction give equation? When alcohol and acid reacts to form ester the reaction is termed as esterification. Activity: Take 1 ml of pure ethanol in a test tube and add 1ml of ethanoic acid to it. Then add 2 to 3 drops of concentrated sulphuric acid to the mixture.
| Reactants | Products | Types of Reactions |
|---|---|---|
| HCl+NaOH | FeS | Combination |
Similarly, what is an esterification reaction?
Esterification is a chemical reaction that forms at least one ester (= a type of compound produced by reaction between acids and alcohols). Esters are produced when acids are heated with alcohols in a process called esterification. An ester can be made by an esterification reaction of a carboxylic acid and an alcohol.
What is the process of esterification?
Esterification is a chemical reaction that forms at least one ester (= a type of compound produced by reaction between acids and alcohols). Esters are produced when acids are heated with alcohols in a process called esterification. An ester can be made by an esterification reaction of a carboxylic acid and an alcohol.
Related Question AnswersWhat are esters made of?
In chemistry, an ester is a chemical compound derived from an acid (organic or inorganic) in which at least one –OH (hydroxyl) group is replaced by an –O–alkyl (alkoxy) group. Usually, esters are derived from a carboxylic acid and an alcohol.What is esterification used for?
The continuous esterification process is used industrially to make methyl acetate from acetic acid and methanol and ethyl acetate from acetic acid and ethanol. The alternative process of making esters from the reaction of an alcohol with an anhydride is important in the manufacture of drugs.Are esters soluble in water?
Solubility in water The small esters are fairly soluble in water but solubility falls with chain length. The reason for the solubility is that although esters can't hydrogen bond with themselves, they can hydrogen bond with water molecules.Is COOH an acid or base?
Carboxyl groups are weak acids, dissociating partially to release hydrogen ions. The carboxyl group (symbolized as COOH) has both a carbonyl and a hydroxyl group attached to the same carbon atom, resulting in new properties.How do you test for esters?
A simple way of detecting the smell of the ester is to pour the mixture into some water in a small beaker. Apart from the very small ones, esters are fairly insoluble in water and tend to form a thin layer on the surface. Excess acid and alcohol both dissolve and are tucked safely away under the ester layer.What does an ester smell like?
Esters. Esters are polar molecules, but their boiling points are lower than those of carboxylic acids and alcohols of similar molecular weight because there is no intermolecular hydrogen bonding between ester molecules. Acids have unpleasant smells, but esters have fruity smells.What ester smells like bananas?
Isoamyl acetate, also known as isopentyl acetate, is an organic compound that is the ester formed from isoamyl alcohol and acetic acid. Isoamyl acetate has a strong odor which is also described as similar to both banana and pear.What is esterification reaction with example?
As a specific example of an esterification reaction, butyl acetate can be made from acetic acid and 1-butanol. A commercially important esterification reaction is condensation polymerization, in which a reaction occurs between a dicarboxylic acid and a dihydric alcohol (diol), with the elimination of water.Is esterification sn1 or sn2?
Organic Mechanisms with Biochem Examples. Fischer esterification involves the formation of an ester from a carboxylic acid and an alcohol. (An alternate method of ester formation involves the use of the carboxylate anion as an SN2 nucleophile upon a primary alkyl halide.)What is the first step of an esterification reaction?
In the first step, the ethanoic acid takes a proton (a hydrogen ion) from the concentrated sulphuric acid. The proton becomes attached to one of the lone pairs on the oxygen which is double-bonded to the carbon.What chemicals are used in esterification?
Esterification- Hydrolysis.
- Acetic Acid.
- Glycerol.
- Ester.
- Ethanol.
- Methanol.
- Hydroxyl Group.
- Distillation.



