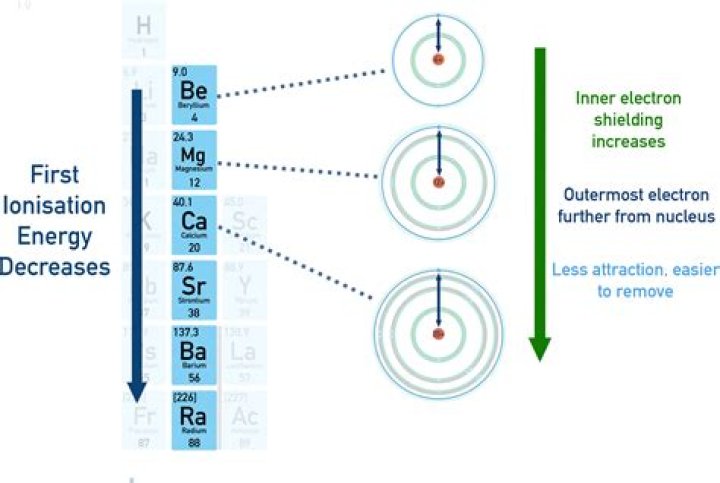
Why does first ionisation decrease down a group?
.
Similarly, you may ask, why the ionization energy decreases down a group?
When moving to the right of a period, the number of electrons increases and the strength of shielding increases. As a result, it is easier for valence shell electrons to ionize, and thus the ionization energy decreases down a group. This is due to electron shielding.
Beside above, why does lattice energy decrease down a group? a) Radius of ions As the size of halide increases down the group, the lattice energy decreases. This is because with the increase of size of ions, the distance between their nuclei increases. Thus the attraction between them decreases and finally the less lattice energy released during the process.
Also to know is, why is the first ionization energy down a group?
Going down a group, the ionisation energy decreases. This is due to the shielding or screen effect of the outer electrons from the nucleus and so the attraction is weaker and they are more easily removed. A comparison of the first ionisation energies of some alkali metals is shown below.
Why does the ionization enthalpy gradually decreases in a group?
Ionization energy decreases as we move down a group because: As we move down, a new full energy level is being added. More electrons means more repulsion. This creates the shielding effect where the addition of the shells, shields the outer electron from receiving the nucleic charge.
Related Question AnswersWhat is the trend in ionization energy down a group?
The general trend is for ionization energy to decrease moving from top to bottom down a periodic table group. Moving down a group, a valence shell is added. The outermost electrons are further from the positive-charged nucleus, so they are easier to remove.Why does ionization energy increase up a group?
The ionization energy of the elements increases as one moves up a given group because the electrons are held in lower-energy orbitals, closer to the nucleus and thus more tightly bound (harder to remove).What happens to electronegativity down a group?
So, as you move down a group on the periodic table, the electronegativity of an element decreases because the increased number of energy levels puts the outer electrons very far away from the pull of the nucleus. Electronegativity increases as you move from left to right across a period on the periodic table.What affects ionization energy?
The ionization energy increases as each electron is removed. Ionization energies are dependent upon the atomic radius. The more electrons shielding the outer electron shell from the nucleus, the less energy required to expel an electron from said atom. The higher the shielding effect the lower the ionization energy.Why does reactivity increase down a group?
These react by losing electrons and reactivity increases as you go down the group. This is because the increased number of electron shells results in more shielding and a greater distance between the outer electrons and the nucleus, which reduces the attraction of the electrons to the nucleus.What is the trend for electronegativity?
The higher the electronegativity of an atom, the greater its ability to attract shared electrons. The electronegativity of atoms increases as you move from left to right across a period in the periodic table. The electronegativity of atoms decreases as you move from top to bottom down a group in the periodic table.Which element has the highest ionization energy?
From this trend, Cesium is said to have the lowest ionization energy and Fluorine is said to have the highest ionization energy (with the exception of Helium and Neon).Why does electronegativity increase?
Electronegativity increases as you move across the periodic table from left to right. This occurs due to a greater charge on the nucleus, causing the electron bonding pairs to be very attracted to atoms placed further right on the periodic table. Fluorine is the most electronegative element.What is the ground state of sodium?
Sodium atoms have 11 electrons and the shell structure is 2.8. 1. The ground state electron configuration of ground state gaseous neutral sodium is [Ne]. 3s1 and the term symbol is 2S1/2.What is the first ionization energy?
Defining first ionisation energy Definition. The first ionisation energy is the energy required to remove one mole of the most loosely held electrons from one mole of gaseous atoms to produce 1 mole of gaseous ions each with a charge of 1+.What is the first ionization energy of magnesium?
Ionisation Energies and electron affinity The electron affinity of magnesium is 0 kJ mol-1.Why are there dips in ionization energy?
Because of the extra orbital, the atomic radii increases, and the electrons are further away from the nucleus. Thus it takes less energy to separate an electron from its nucleus. The extra orbital has its electron density farther away from the nucleus, and hence the slight drop in ionization energy.What is the ionization energy of nitrogen?
The ionization energy of molecular nitrogen is 1503 kJ mol?-1, and that of atomic nitrogen is 1402 kJ mol?-1. Once again, the energy of the electrons in molecular nitrogen is lower than that of the electrons in the separated atoms, so the molecule is bound.What is the first ionization energy of lithium?
you stated that the first ionization energy for Li is 520 kj/mol and the second ionization energy is 7298kj/mol.What does electronegativity mean?
Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. The Pauling scale is the most commonly used. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.Does Lattice Energy increase down a group?
WHY? - The atomic radius increases as you move down a group. Since the square of the distance is inversely proportional to the force of attraction, lattice energy decreases as the atomic radius increases. Therefore, lattice energy increases as the charges increase.How do you solve for lattice energy?
You should talk about "lattice dissociation enthalpy" if you want to talk about the amount of energy needed to split up a lattice into its scattered gaseous ions. For NaCl, the lattice dissociation enthalpy is +787 kJ mol-1.| kJ | |
|---|---|
| electron affinity of Cl (x 2) | -698 |
| lattice enthalpy | -2526 |
| calculated ΔHf | -643 |