And CaCl2 is a better road salt than NaCl because it makes more ions per formula unit of the salt dissolved..
Likewise, people ask, why is calcium chloride used to salt roads instead of sodium chloride?
Calcium chloride is an effective deicer, working at temperatures below most competing products, and is significantly more effective than sodium chloride because of its ability to extract moisture from its surroundings and to cause exothermic or heat generating reactions.
Also Know, what is the benefit of using a combination of salt and calcium chloride to salt the roads over using salt alone? Calcium chloride: Calcium chloride has several advantages over salt. It also works by reducing the freezing point of water, but it's effective down to minus 25 degrees F (minus 31 C), while salt only works to about 15 F (minus 9 C).
Furthermore, which salt NaCl or CaCl2 has a greater effect on freezing point?
For the same molar concentration of NaCl and CaCl2, CaCl2 would be more effective in lowering freezing point. This is because freezing point depression is dependent on the number of ions present (colligative property). The more ions present in a solution, the greater the change in temperature.
Is calcium chloride more corrosive than salt?
Calcium Chloride (Pellets) It is much more costly than rock salt. It only damages concrete slightly. Has a minor effect on plants, but is very corrosive to metals. It will melt ice at up to minus 20 degrees Fahrenheit.
Related Question Answers
What is the common name for sodium chloride?
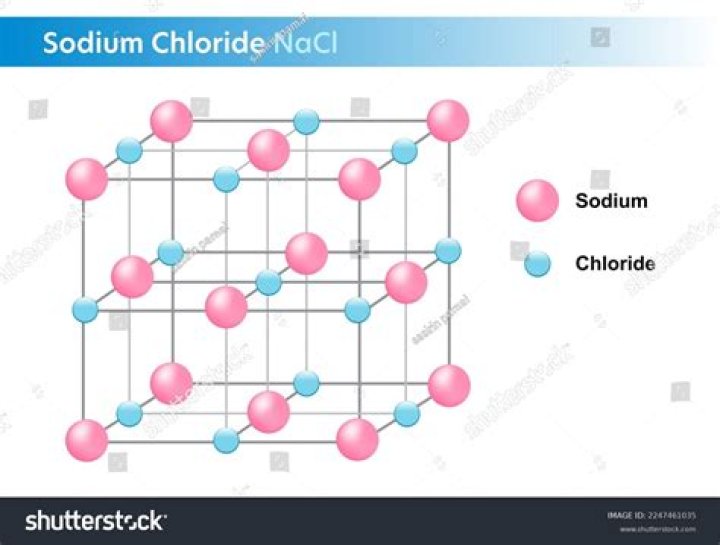
Sodium chloride /ˌso?di?m ˈkl?ːra?d/, commonly known as salt (though sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions.Why is cacl2 better than NaCl at melting ice?
Calcium chloride is more effective at melting ice because it can break down into three ions instead of two: one calcium ion and two chloride ions. While some other compounds that can melt ice and snow don't include chloride, they are much more expensive than sodium chloride or calcium chloride.What are the benefits of calcium chloride?
Calcium Chloride is a mineral indicated in the immediate treatment of hypocalcemic tetany (abnormally low levels of calcium in the body that cause muscle spasm). Calcium chloride injection is also used in cardiac resuscitation, arrhythmias, hypermagnesemia, calcium channel blocker overdose, and beta-blocker overdose.What can I use instead of calcium chloride?
Alum is a substitute for calcium chloride. Alum is used to crisp cucumbers during pickling process. Also, rock salt can be used in place of calcium chloride.Can salt melt?
Depending on what type of salt (like not table salt), you can melt it more easily than metals. Table salt, however, has a melting temperature of 801 degrees C. Like many other properties, that's less than steel.Is calcium chloride toxic?
If ingested, calcium chloride can lead to burns in the mouth and throat, excess thirst, vomiting, stomach pain, low blood pressure, and other possible severe health effects. In extreme cases of exposure or ingestion, this chemical can cause skin burns, cardiac disturbances, respiration issues, and seizures.What happens when you mix water and calcium chloride?
Calcium chloride produces heat (exothermic) when it dissolves in water, while sodium bicarbonate absorbs heat (endothermic) as it dissolves. Calcium chloride, baking soda, and water combine to produce carbon dioxide gas.What products have calcium chloride?
As a firming agent, calcium chloride is used in canned vegetables, in firming soybean curds into tofu and in producing a caviar substitute from vegetable or fruit juices. It is commonly used as an electrolyte in sports drinks and other beverages, including bottled water.Why is CaCl2 more soluble than NaCl?
A few water molecules do not have enough strength to get in between and dissolve it. Chloride is less electronegative, thus CaCl2 has weaker bonds, So weaker the bond strength, greater the solubility – at least that's the expected. Hence, CaCl2 is more soluble than CaF2.What is the difference between NaCl and CaCl2?
The key difference between calcium chloride and sodium chloride is that calcium chloride molecule has two chlorine atoms whereas sodium chloride molecule has one chlorine atom. The chemical formula of calcium chloride is CaCl2. The chemical formula of sodium chloride is NaCl.Which solution of salt NaCl or CaCl2 is better to use on icy roads?
When we spread CaCl2 on roads and walkways for safety in the winter, we are taking advantage of the freezing point depression idea. And CaCl2 is a better road salt than NaCl because it makes more ions per formula unit of the salt dissolved.What temperature does NaCl freeze?
801 °C
Is salt bad for roads?
Rock salt helps keep roads safe when winter storms hit, reducing winter road accidents. But it can also have serious, negative effects on aquatic ecosystems. At high concentrations, salt can be fatal to some aquatic animals. This rock salt is similar to table salt, made up of sodium and chloride, but coarser.Why do we salt roads?
The biggest reason for pouring salt on icy roads is that salt lowers the freezing point of water. Water normally freezes at 32 degrees Fahrenheit, but when you add salt, that threshold drops to 20 degrees (or even 2 degrees) Fahrenheit. You can watch as the water surrounding each grain of salt melts.What type of salt is used on roads?
Sodium chloride
What state uses the most road salt?
States in the salt belt include Connecticut, Delaware, Illinois, Indiana, Iowa, Kansas, Maine, Maryland, Massachusetts, Michigan, Minnesota, Missouri, Nebraska, New Hampshire, New Jersey, New York, North Dakota, Ohio, Pennsylvania, Rhode Island, Vermont, Virginia, West Virginia, Wisconsin, and Washington DC.Is salt bad for concrete?
Salt does not damage concrete, but the effects of salt can. Salt does not chemically react with hardened concrete. Salt does however lower the freezing point of water, attract moisture, and increase pressure of frozen water.What is road salt made of?
Road salt is halite, which is the natural mined mineral form of table salt or sodium chloride (NaCl). While table salt has been purified, rock salt contains mineral impurities, so it is typically brownish or gray in color.