When NaCl is added to water what is added to table salt
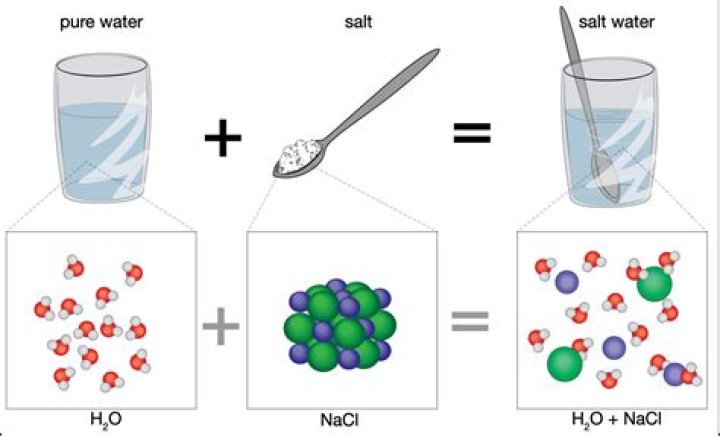
When table salt is placed in water, the slightly electropositive sodium portion is attracted to the slightly electronegative oxygen portion of water molecules. At the same time, the slightly electronegative chlorine portion of NaCl is attracted to the slightly electropositive hydrogen portion of water.
What happens when table salt NaCl is added to water?
When salt is mixed with water, the salt dissolves because the covalent bonds of water are stronger than the ionic bonds in the salt molecules. … Water molecules pull the sodium and chloride ions apart, breaking the ionic bond that held them together.
What happens when you dissolve NaCl in water quizlet?
Dissolving happens when the attractions between the water molecules and the sodium chloride ions overcome the attraction of the ions to each other, causing the ions to separate from each other and mix into the water.
What is the mixture of table salt NaCl?
Sodium chloride , commonly known as salt (although sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. With molar masses of 22.99 and 35.45 g/mol respectively, 100 g of NaCl contains 39.34 g Na and 60.66 g Cl.Why is NaCl soluble in water?
Water can dissolve salt because the negative chloride ions are attracted by the positive portion of water molecules and the positive sodium ions are attracted by the negative portion of water molecules. …
What is added to salt?
Iodine is a micronutrient, which means we need it in small quantities to be healthy. Because iodine is relatively rare in many people’s normal diets, it’s added to table salt. Then when people salt their food, such as tasty turkey, stuffing and mashed potatoes, they’re also getting iodine.
Is NaCl a compound or mixture?
Salt is another compound. Its chemical formula is NaCl, which stands for sodium chloride.
When salt NaCl is dissolving in water H2O what happens to the attraction between the salt ions and the oxygen atoms of the water?
Dissolving happens when the attractions between the water molecules and the sodium and chloride ions overcome the attractions of the ions to each other. This causes the ions to separate from one another and become thoroughly mixed into the water.What is the name of the process that occurs when NaCl is placed in water?
1: The dissolving of sodium chloride in water. After coming apart from the crystal, the individual ions are then surrounded by solvent particles in a process called solvation. Note that the individual Na+ ions are surrounded by water molecules with the oxygen atom oriented near the positive ion.
When table salt is dissolved in water the sodium and chloride ions quizlet?Terms in this set (29) When table salt is dissolved in water, the sodium and chloride ions: dissociate.
Article first time published onHow does NaCl dissolve in water?
Just dump all the solid NaCl into the water, put a stir bar and stir until it all dissolves. Dissolution of NaCl may be slow when getting closer to saturation, but it should all dissolve.
How does salt draw out water?
Answer: Technically, salt draws out moisture through the process of osmosis. This is the basis for all the theories about drying and toughening properties of salt when in contact with foods.
What type of mixture is salt dissolved in water?
The salt dissolved in water is a homogeneous mixture, or a solution (Figure 3.5.
Is table salt a mixture?
Mixtures. Ordinary table salt is called sodium chloride. It is considered a pure substance because it has a uniform and definite composition. … A mixture is a physical blend of two or more components, each of which retains its own identity and properties.
Is NaCl a homogeneous mixture?
(This qualifies as a mixture, since evaporation allows reformation of sodium chloride solid again, thus separating the components, NaCl and H2O , via physical means. It is known as a homogeneous mixture.)
Is table salt a compound?
In chemical terms, salts are ionic compounds. To most people, salt refers to table salt, which is sodium chloride. Sodium chloride forms from the ionic bonding of sodium ions and chloride ions. There is one sodium cation (Na+) for every chloride anion (Cl–), so the chemical formula is NaCl (Fig.
Is table salt iodized?
Iodized salt is salt that contains small amounts of sodium iodide or potassium iodide. It’s normal salt that has been sprayed with potassium iodate. It looks and tastes the same! The majority of table salt used nowadays is iodized, and it comes with many benefits.
Why does table salt have potassium iodide added to it?
That is why the carton or bag identifies the product as “iodized” salt. Why is this done? It is to make sure that people get enough iodine in their diet, to prevent disease of the thyroid gland.
How do they iodized salt?
Iodine is added as potassium iodate to salt after refining and drying and before packing. Iodization can often be linked with existing production and/or refining lines. This can be done by adding a solution of potassium iodate to the salt or by adding dry potassium iodate powder.
When NaCl is added to water what is the boiling point?
If you add salt to water, you raise the water’s boiling point, or the temperature at which it will boil. The temperature needed to boil will increase about 0.5 C for every 58 grams of dissolved salt per kilogram of water. This is an example of boiling point elevation, and it is not exclusive to water.
What change is salt dissolving in water?
For example salt dissolving in water is usually considered to be a physical change, however the chemical species in salt solution (hydrated sodium and chlorine ions) are different from the species in solid salt.
Is NaCl ionic or covalent?
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons.
When salt is dissolved in water water is the quizlet?
Terms in this set (10) When you dissolve salt in water, which compound is the solute and which is the solvent? The solute is what you are dissolving, and the solvent is what you are dissolving into. Therefore, salt is the solute, and water is the solvent.
When salt dissolves in water what type of substance is formed quizlet?
Salt dissolves in water to form a homogeneous mixture of saltwater. You just studied 20 terms!
When placed into water what is the ionic compound NaCl?
When an ionic compound, or salt, (e.g. NaCl) is placed in water, its ionic components (e.g. Na+ and Cl-) dissociate (separate) and water molecules surround each ion. This is shown in figure 2.3.
How do you dissolve table salt?
Take 3 glasses of water. Add cold water to one (either ice water, or water from the fridge), add room temperature water to the second, and add hot water to the third. Then, add 1 teaspoon of salt to each glass at the same time, and time how long it takes each to dissolve.
Is NaCl dissolving in water endothermic or exothermic?
Dissolution of NaCl in waterDissolution of sodium chloride in water is endothermic. Solute-solvent attractive bond formation (the exothermic step in the process of solvation) is indicated by dashed lines.
Does salt release water?
When you add the salt, vegetable release stores water, this is happening due to the osmosis (active transport) process. Actually when we add salt the concentration of the salt outside the vegetable increases which leads to the flow of water molecules from the vegetable.
Why does salt attract water?
Unlike pepper, table salt is hygroscopic, meaning that because of the net positive charge of its chemical components, or ions, it can attract atmospheric water, which has a net negative charge. Traces of salt atop the shaker may attract visible water.
How is salt related to chemistry?
salt, in chemistry, substance produced by the reaction of an acid with a base. A salt consists of the positive ion (cation) of a base and the negative ion (anion) of an acid. The reaction between an acid and a base is called a neutralization reaction.
How are mixture formed if salt dissolve in water?
Electrostatic attraction is the force that causes this event. After this process, water molecules get between sodium and chlorine ions and separate them. Thus the salt is dissolved in water. The isolated ions are spread evenly in the solvent to form a homogeneous mixture.



