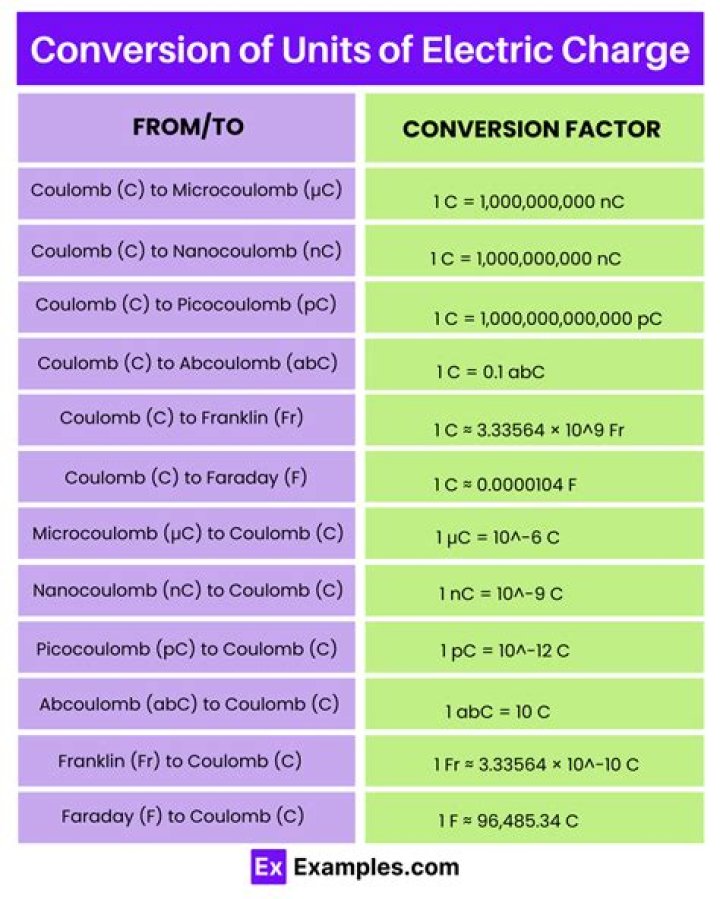
The mole (symbol: mol) is the unit of measurement for amount of substance in the International System of Units (SI). The term gram-molecule (g mol) was formerly used for "mole of molecules", and gram-atom (g atom) for "mole of atoms". For example, 1 mole of MgBr2 is 1 gram-molecule of MgBr2 but 3 gram-atoms of MgBr2..
Regarding this, what is the SI unit for mole?
The mole, symbol mol, is the SI unit of amount of substance. One mole contains exactly 6.022 140 76 x 1023 elementary entities. This number is the fixed numerical value of the Avogadro constant, NA, when expressed in the unit mol–1 and is called the Avogadro number.
Secondly, what is the mole in chemistry? The mole is the unit of amount in chemistry. A mole of a substance is defined as: The mass of substance containing the same number of fundamental units as there are atoms in exactly 12.000 g of 12C. Fundamental units may be atoms, molecules, or formula units, depending on the substance concerned.
Also, what are some units for moles?
The mole, abbreviated mol, is an SI unit which measures the number of particles in a specific substance. One mole is equal to 6.02214179×1023 atoms, or other elementary units such as molecules.
Why is a mole 6.022 x10 23?
The mole allows scientists to calculate the number of elementary entities (usually atoms or molecules) in a certain mass of a given substance. Avogadro's number is an absolute number: there are 6.022×1023 elementary entities in 1 mole. This can also be written as 6.022×1023 mol-1.
Related Question Answers
How many moles are in a gram?
The answer is 0.0087094358027487. We assume you are converting between moles In and gram. You can view more details on each measurement unit: molecular weight of In or grams The SI base unit for amount of substance is the mole. 1 mole is equal to 1 moles In, or 114.818 grams.What is a mole in units?
Mole (unit) The mole (symbol: mol) is the base unit of amount of substance ("number of substance") in the International System of Units or System International (SI), defined as exactly 6.02214076×1023 particles, e.g., atoms, molecules, ions or electrons.What is meant by 1 mole?
One mole is defined as the amount of substance containing as many elementary entities (atoms, molecules, ions, electrons, radicals, etc.) as there are atoms in 12 grams of carbon-12 (6.023 x 1023). The mass of one mole of a substance equals its relative molecular mass expressed in grams. Also spelled as mol.What are the 7 basic units?
There are seven base units in the SI system: - the kilogram (kg), for mass.
- the second (s), for time.
- the kelvin (K), for temperature.
- the ampere (A), for electric current.
- the mole (mol), for the amount of a substance.
- the candela (cd), for luminous intensity.
- the meter (m), for distance.
Who discovered the mole?
Amadeo Avogadro
What is a mole in physics?
The mole is the SI unit for the amount of a substance and one of the seven fundamental SI units. It is defined as the amount of substance of a system that contains as many elementary entities as there are atoms in 0.012 kilograms of carbon-12 (BIPM 1998, p. 97).What causes moles?
What Causes a Mole? Moles occur when cells in the skin grow in a cluster instead of being spread throughout the skin. These cells are called melanocytes, and they make the pigment that gives skin its natural color. Moles may darken after exposure to the sun, during the teen years, and during pregnancy.How do I calculate moles?
Use the molecular formula to find the molar mass; to obtain the number of moles, divide the mass of compound by the molar mass of the compound expressed in grams.How big is a mol?
Mole, also spelled mol, in chemistry, a standard scientific unit for measuring large quantities of very small entities such as atoms, molecules, or other specified particles. The mole designates an extremely large number of units, 6.02214076 × 1023.What is the symbol of mole in math?
As of 2017 as defined by IUPAC, the mole, symbol mol, is the SI unit of amount of substance. One mole contains exactly 6.022 140 76 × 10 elementary entities. This number is the fixed numerical value of the Avogadro constant, when expressed in mol and is called the Avogadro number.How much is a mole of water?
The average mass of one mole of H2O is 18.02 grams. This is stated: the molar mass of water is 18.02 g/mol.Why is mole concept important?
The mole is important because it allows chemists to work with the subatomic world with macro world units and amounts. Atoms, molecules and formula units are very small and very difficult to work with usually. Defining the mole in this way allows you change grams to moles or moles to particles.How many particles are in a mole?
In science, we have a name for this, called Avogadro's number, and it describes the number of representative particles in one mole of a substance. The inverse mole unit tells us there are 6.022×1023 particles of something *per mole*.What is Mole in Chemistry with example?
A mole corresponds to the mass of a substance that contains 6.023 x 1023 particles of the substance. The mole is the SI unit for the amount of a substance. Its symbol is mol. By definition: 1 mol of carbon-12 has a mass of 12 grams and contains 6.022140857 x 1023 of carbon atoms (to 10 significant figures). Examples.Do moles have eyes?
Moles. Moles are small, burrowing mammals. Their eyes are poorly developed, but what they lack in sight, they make up for in their sense of touch. All moles have very sensitive snouts and long, clawed digits that they use to dig tunnels.How many moles are in 64 grams of ch4?
16*4= 64. 4 moles of methane have 64g. Also, 1 mole has 6*10 raised to 23 molecules.What is a group of moles called?
Male moles are called "boars", females are called "sows". A group of moles is called a "labour".