Cyanohydrins have the structural formula of R2C(OH)CN. The “R” on the formula represents an alkyl, aryl, or hydrogen. In order to form a cyanohydrin, a hydrogen cyanide adds reversibly to the carbonyl group of an organic compound thus forming a hydroxyalkanenitrile adducts (commonly known and called as cyanohydrins)..
Likewise, what is a Cyanohydrin molecule?
A cyanohydrin is an organic compound that contains both a cyanide and a hydroxy group on an aliphatic section of the molecule. Because cyanohydrins are primarily used as chemical intermediates, data on production and prices are not usually published.
Also, what happens when acetaldehyde reacts with HCN? When acetaldehyde react with HCN, cyanohydrin(CH3CH(OH)CN) will form and when this is subjected to acid hydrolysis an alpha hydroxy acid(CH3CH(OH)COOH) or alpha-beta unsaturated acid(CH2=CH(OH)COOH)will form.
Similarly, you may ask, what is Cyanohydrin formation?
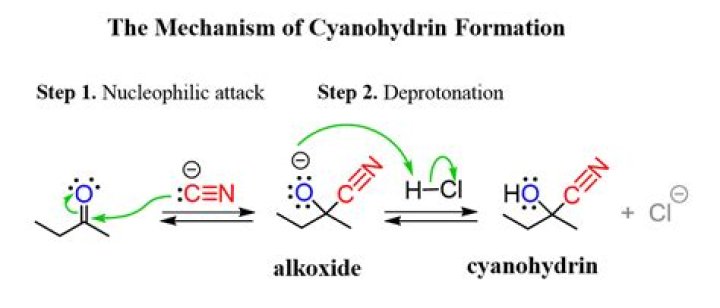
A cyanohydrin reaction is an organic chemical reaction by an aldehyde or ketone with a cyanide anion or a nitrile to form a cyanohydrin. This nucleophilic addition is a reversible reaction but with aliphatic carbonyl compounds equilibrium is in favor of the reaction products.
What is meant by Semicarbazone?
In organic chemistry, a semicarbazone is a derivative of imines formed by a condensation reaction between a ketone or aldehyde and semicarbazide.
Related Question Answers
How do you convert cyanide to aldehyde?
The aluminum atom in DIBAL acts as a Lewis acid, accepting an electron pair from the nitrile. The nitrile is then reduced by the transfer of a hydride ion to the carbon of the carbon-nitrile triple bond, producing an imine. After a workup with water, the aluminum complex is hydrolyzed to produce the desired aldehyde.What is NACN in organic chemistry?
It has a role as an EC 1.15. 1.1 (superoxide dismutase) inhibitor. It is a cyanide salt, a one-carbon compound and a sodium salt. Cyanide is usually found joined with other chemicals to form compounds.How does HCN react?
Aldehydes and ketones undergo reaction with HCN to produce cyanohydrins. Hence, base as a catalyst helps to speed up the reaction. This is because catalysis helps in the generation of cyanide ion (CN) which acts as a stronger nucleophile and adds to carbonyl compounds to produce the corresponding cyanohydrin.Why is potassium cyanide used instead of hydrogen cyanide?
The reaction isn't normally done using hydrogen cyanide itself, because this is an extremely poisonous gas. Instead, the aldehyde or ketone is mixed with a solution of sodium or potassium cyanide in water to which a little sulphuric acid has been added. The rate of the reaction falls if the pH is any higher.What type of reaction takes place upon treatment of a ketone with HCN to form a Cyanohydrin?
What type of reaction takes place upon treatment of a ketone with HCN to form a cyanohydrin? Explanation: The atoms of HCN add to the carbon-oxygen double bond of the ketone by nucleophilic attack of the cyanide anion on the electrophilic carbon of the carbonyl.How do you make acetal?
Formation of an acetal occurs when the hydroxyl group of a hemiacetal becomes protonated and is lost as water. The carbocation that is produced is then rapidly attacked by a molecule of alcohol. Loss of the proton from the attached alcohol gives the acetal.Is HCN a strong acid?
HCN, also known as hydrocyanic acid or prussic acid, is a weak acid. According to this source, the of HCN is , which is really tiny. Memorize the short list of acids that are typically thought of as strong. This includes HCl, HBr, HI, HNO3, HClO3, HClO4, and H2SO4.Why is nucleophilic addition important in organic synthesis?
The reactions are involved in the biological synthesis of compounds in the metabolism of every living organism, and are used by chemists in academia and industries such as pharmaceuticals to prepare most new complex organic chemicals, and so are central to organic chemistry.Are imines basic?
Acid-base reactions Somewhat like the parent amines, imines are mildly basic and reversibly protonate to give iminium salts. Since imines derived from unsymmetrical ketones are prochiral, their reduction is a useful method for the synthesis of chiral amines.Is HCN a strong Nucleophile?
The cyanide ion as a nucleophile All nucleophiles contain an active lone pair of electrons. The cyanide ion comes from hydrogen cyanide, which is a covalent molecule. Hydrogen cyanide is very weakly acidic, which means that it can lose a hydrogen ion - although not very easily.What is prussic acid used for?
… cyanide in water is called hydrocyanic acid, or prussic acid. It was discovered in 1782 by a Swedish chemist, Carl Wilhelm Scheele, who prepared it from the pigment Prussian blue. Hydrogen cyanide and its compounds are used for many chemical processes, including fumigation, the case hardening of iron and steel,…What does KCN do in a reaction?
The facts of the reaction are exactly the same as with primary halogenoalkanes. If the halogenoalkane is heated under reflux with a solution of sodium or potassium cyanide in ethanol, the halogen is replaced by -CN, and a nitrile is produced.What is aldol condensation write its mechanism?
Aldol Condensation can be defined as an organic reaction in which enolate ion reacts with a carbonyl compound to form β-hydroxy ketone or β-hydroxy aldehyde, followed by dehydration to give a conjugated enone. Aldol Condensation plays a vital role in organic synthesis, creating a path to form carbon-carbon bonds.Where is cyanide from Wikipedia?
Cyanides are produced by certain bacteria, fungi, and algae and are found in a number of plants. Cyanides are found in substantial amounts in certain seeds and fruit stones, e.g., those of bitter almonds, apricots, apples, and peaches. Chemical compounds that can release cyanide are known as cyanogenic compounds.Which of the aldehyde is most reactive?
Yes, formaldehyde is the most reactive almost all the aldehydes cause is no methylene grp attached to it. As the aldehyde increases in length there are more bulky group attached which decreses the positive inductive effect on the carbon of the aldehydic grp.Is cyanide a nucleophile?
Cyanide ions as nucleophiles Nucleophiles are either fully negative ions, or else have a strongly - charge somewhere on a molecule. In the case of the cyanide ion, there is a full negative charge on the carbon, as well as a lone pair of electrons.What compounds react with hydrogen cyanide?
Aldehydes and Ketones: Nucleophilic Addition Reactions Hydrogen cyanide adds to an aldehyde or ketone to give a compound called a cyanohydrin. In the product, the nucleophilic cyano group is bonded to the carbonyl carbon, and the proton is bonded to the carbonyl oxygen.Why is HCN a weak acid?
Hydrogen cyanide, or HCN, is not a strong acid. Hydrogen cyanide has a pKa of 9.2. The carbon has a triple bond with the nitrogen and a single bond with hydrogen. It is a weak acid because the carbon is reluctant to donate the hydrogen atom because the bond keeps the molecule stable.Why is ch3cho more reactive than c6h5cho towards reaction with HCN?
electrophilic substitution reaction due to its partial double bond character.. The carbon atom of cabonyl group of C6H5CHO is less electrophillic than the carbon atom of carbonyl group of CH3CHO. Hence CH3CHO being more electropositive is more reactive towards nucleophilic addition of HCN.