What is the principle of Charles Law
The physical principle known as Charles’ law states that the volume of a gas equals a constant value multiplied by its temperature as measured on the Kelvin scale (zero Kelvin corresponds to -273.15 degrees Celsius).
How does Charles's law work?
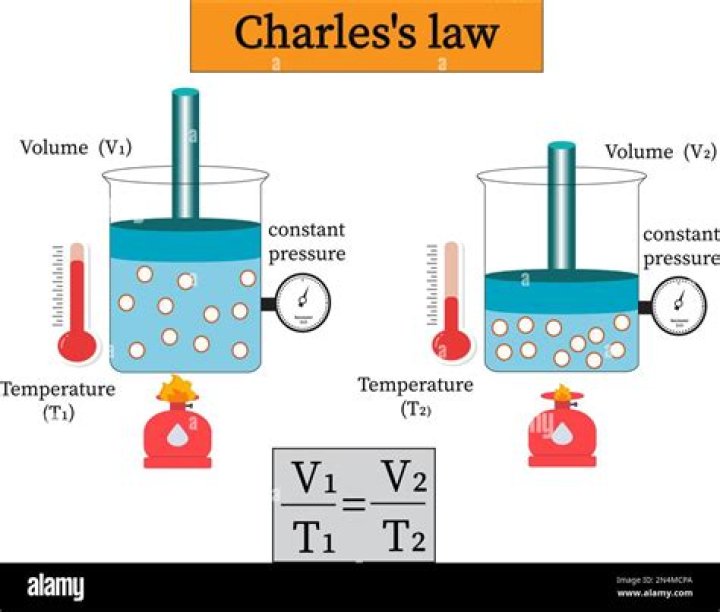
Charles’ Law is an experimental gas law that describes how gases tend to expand when heated. The law states that if a quantity of gas is held at a constant pressure, there is a direct relationship between its volume and the temperature, as measured in degrees Kelvin.
Why is Charles's law true?
Increasing the temperature of a volume of gas causes individual gas molecules to move faster. As the molecules move faster, they encounter the walls of the container more often and with more force. … Charles’ Law is the formal description of this relationship between temperature and volume at a fixed pressure.
What is Boyle's Law and Charles Law?
Boyle’s law—named for Robert Boyle—states that, at constant temperature, the pressure P of a gas varies inversely with its volume V, or PV = k, where k is a constant. … Charles (1746–1823)—states that, at constant pressure, the volume V of a gas is directly proportional to its absolute (Kelvin) temperature T, or V/T = k.Why do we use Charles Law?
Charles’s law defines the direct relationship between temperature and volume. When the parameters of a system change, Charles’s law helps us anticipate the effect the changes have on volume and temperature.
What does Boyles law state?
This empirical relation, formulated by the physicist Robert Boyle in 1662, states that the pressure (p) of a given quantity of gas varies inversely with its volume (v) at constant temperature; i.e., in equation form, pv = k, a constant. …
What's an example of Charles Law?
One easy example of Charles’ Law is a helium balloon. If you fill a helium balloon in a warm or hot room, and then take it into a cold room, it shrinks up and looks like it has lost some of the air inside. But if you take it back to a warm or hot place, it fills back up and seems to be full again.
What kind of relationship is Charles Law?
Charles Law states that the volume of a given mass of a gas is directly proportional to its Kevin temperature at constant pressure. In mathematical terms, the relationship between temperature and volume is expressed as V1/T1=V2/T2.Why is Boyle's law true?
According to Boyle’s Law, an inverse relationship exists between pressure and volume. Boyle’s Law holds true only if the number of molecules (n) and the temperature (T) are both constant.
Who discovered Charles Law?Jacques CharlesBornNovember 12, 1746 Beaugency, OrléanaisDiedApril 7, 1823 (aged 76) Paris, FranceNationalityFrenchKnown forCharles’s law
Article first time published onHow is Charles law used in everyday life?
Hot Air Balloon You might have wondered about the working of the hot air balloon. Charle’s Law describes that temperature and volume are directly proportional to each other. When a gas is heated, it expands. As the expansion of the gas takes place, it becomes less dense and the balloon is lifted in the air.
How do you prove Charles Law?
The equation for Charles’s law can be expressed as V1/T1=V2/T2. In other words, if a balloon is filled with air, it will shrink if cooled and expand if heated. This happens because the air inside the balloon, which is a gas, takes up a smaller volume when it is cool, and takes up a larger volume when it is heated.
What is the final pressure of the gas?
Gas LawsPressure or Volume Constant Temperature; TOriginal pressure x Original Volume=Final pressure x Final VolumeP1V1=P2V2 IsothermicPressure or temperature Constant Volume: VOriginal Pressure x Final Temperature=Final Pressure x Original TemperatureP1T2=P2T1 Isochoric
What are the three gas laws?
The gas laws consist of three primary laws: Charles’ Law, Boyle’s Law and Avogadro’s Law (all of which will later combine into the General Gas Equation and Ideal Gas Law).
What is K in Boyle's law?
Simply put, Boyle’s states that for a gas at constant temperature, pressure multiplied by volume is a constant value. The equation for this is PV = k, where k is a constant. At a constant temperature, if you increase the pressure of a gas, its volume decreases. … Boyle’s law is a form of the Ideal Gas Law.
Is Charles Law indirect or direct?
Charles Law is a direct relationship between temperature and volume. When the temperature of the molecules increases the molecules move faster creating more pressure on the container of the gas increasing the volume, if the pressure remains constant and the number of the molecules remains constant.
What did Charles Law discovery?
Charles’ Law: The Volume Is Directly Proportional to Temperature. In 1787, French chemist Jacques Charles was experimenting on the relationship between the volume and temperature of a gas. What he found was that, if he kept the pressure constant, that the volume of a gas was proportional to the gas’s temperature.
When was Charles's law created?
Quantitative experiments establishing the law were first published in 1802 by Gay-Lussac, who credited Jacques Charles with having discovered the law earlier. Charles’ law relates the volume and temperature of a gas when measurements are made at constant pressure.
How does Charles Law relate to breathing?
As the air warms it expands to a larger volume. Charles’ Law does not affect breathing nearly as much as Boyle’s Law does, but it does have an effect. … If it is a cold day with a temperature of -10 °C, the inhaled air will expand as it reaches the temperature in your lungs (37 °C).
What is Charles law and its derivation?
Charles’ Law states that at constant pressure, the volume of a fixed mass of a dry gas is directly proportional to its absolute temperature. We can represent this using the following equation: VαT. Since V and T vary directly, we can equate them by making use of a constant k.
What is the conclusion of Charles Law?
Charles Law states that “as temperature increases, so does the volume of a gas sample when the pressure is held constant”. The result of V1/T1 and V2/T2 were very close to each other. This is due to the fact that this experiment was done in a closed system.
What is V2 in chemistry?
You can solve for the concentration or volume of the concentrated or dilute solution using the equation: M1V1 = M2V2, where M1 is the concentration in molarity (moles/Liters) of the concentrated solution, V2 is the volume of the concentrated solution, M2 is the concentration in molarity of the dilute solution (after …
What units are used in Charles Law?
Note: Charles’s Law uses kelvin for temperature units, while the volume units can be any volume unit (mL, L, etc.)
What is the final volume of gas?
Assume that the pressure and the amount of the gas remain constant. Solving for the final volume, V f = 22.7 L. So, as the temperature is increased, the volume increases. This makes sense because volume is directly proportional to the absolute temperature (as long as the pressure and the amount of the remain constant).
What is the formula for solving final pressure?
p/T = constant, or. p1/p2 = T1/T2 for conditions changing from 1 (initial) to 2 (final), or p1/T1 = p2/T2 for constant volume.
What is STP in chemistry?
Definition. Standard Temperature and Pressure (STP) is defined as 0 degrees Celsius and 1 atmosphere of pressure.



