What is photoelectron spectroscopy used for
Photoelectron spectroscopy (PES) is an experimental technique used to determine the relative energies of electrons in atoms and molecules.
What are the main applications of XPS?
XPS is routinely used to analyze inorganic compounds, metal alloys, semiconductors, polymers, elements, catalysts, glasses, ceramics, paints, papers, inks, woods, plant parts, make-up, teeth, bones, medical implants, bio-materials, coatings, viscous oils, glues, ion-modified materials and many others.
Why is XPS so surface sensitive?
The peak shape and precise position indicates the chemical state for the element. XPS is a surface sensitive technique because only those electrons generated near the surface escape and are detected. The photoelectrons of interest have relatively low kinetic energy.
What can XPS detect?
XPS reveals which chemical elements are present at the surface and the nature of the chemical bond that exists between these elements. It can detect all of the elements except hydrogen and helium.Which radiation is used in XPS studies?
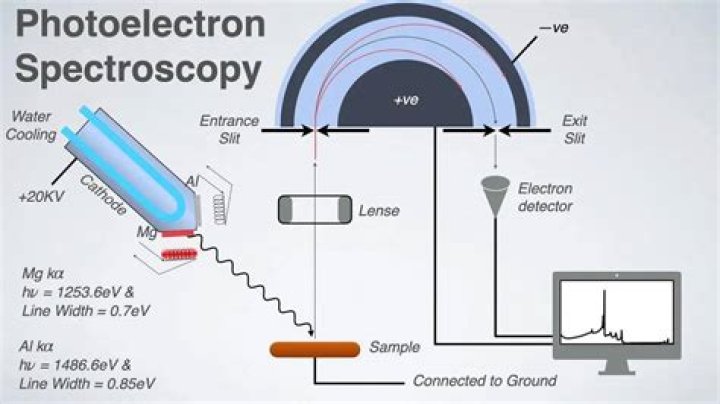
1), satellite peaks, and multiple splitting can be used to identify chemical states. XPS is initiated by irradiating a sample with monoenergetic soft X-rays, most commonly Mg Kα (1253.6 eV with a line width ≈ 0.7 eV) or Al Kα (1486.6 eV with a line width ≈ 0.85 eV).
Can XPS detect H?
Hydrogen and helium are essentially impossible to detect by a lab-based XPS.
Why is XPS useful as a characterization tool?
X-ray photoelectron spectroscopy (XPS) is one of the most powerful tools to characterize thin films materials. … The XPS analyses of oxysulfides thin films at different stages of their cycling process have shown apparently good efficiency of the oxygen-rich compositions.
What is chemical shift in XPS?
Chemical shift: change in binding energy of a core electron of an element due to a change in the chemical bonding of that element.How does Auger electron spectroscopy work?
In Auger electron spectroscopy (AES), we bombard a sample surface with a focused beam of high-energy (2- to 10-kV) electrons. The incident electrons lose energy to the sample atoms, generating Auger electrons that have discrete kinetic energies characteristic of the emitting atoms.
How do you explain XPS data?The Analysis In order to interpret XPS or ESCA results, it’s helpful to understand how this technique actually works. Simply put, XPS uses an x-ray beam to excite atoms on the surface of a solid sample, which spurs the release of photoelectrons.
Article first time published onIs XPS destructive?
XPS is a non-destructive technique to measure surface chemistry of solid materials, in particular the chemical composition and electronic state.
What is the difference between XPS and EDX?
EDX is an analytical technique used for the elemental analysis or chemical characterization of a sample, On the other hand, XPS measures the kinetic energy and number of electrons that escape from the top 1 to 10 nm of the material being analyzed.
What is the difference between UPS and XPS?
XPS uses high energy X-ray photons to excite “core” electrons in the near-surface region UPS uses lower energy photons in the deep UV region to excite valence electrons.
Why XPS is also called ESCA?
XPS requires ultra-high vacuum (UHV) conditions. … XPS is also known as ESCA, an abbreviation for “electron spectroscopy for chemical analysis” introduced by Kai Siegbahn and his research group. Detection limits for most of the elements are in the parts per thousand range (1,000 PPM).
What is Survey spectra in XPS?
High resolution C1s spectrum showing Carbon-Oxygen and Fluorocarbon bonding states. X-ray photoelectron spectroscopy (XPS) is a highly surface-specific chemical analytical technique used to probe the elemental composition and bonding states in the outermost 2-10 nm of a solid surface.
Can XPS detect fluorine?
The elemental composition and chemical bonding are characterized by X-ray photoelectron spectroscopy (XPS). … The result showed XPS could detect the prepared re-deposition contained fluorine 31.2 At%, oxygen 23.7 At%, carbon 23.7 At%, aluminium 16.0 At% and titanium 5.4 At%.
Why is AES surface sensitive?
Surface sensitivity in AES arises from the fact that emitted electrons usually have energies ranging from 50 eV to 3 keV and at these values, electrons have a short mean free path in a solid.
What phenomena occur in Auger spectroscopy?
2 Auger Electron Spectroscopy. In AES, Auger electrons are emitted by a three-body process in which a core electron is ionized by the incoming radiation, a higher-energy electron drops into the vacant state and in doing so releases enough energy to ionize another electron that has an energy characteristic of the atom.
Who invented Auger Electron Spectroscopy?
Historically, the Auger effect was discovered independently in 1922 by L. Meitner [1], an Austrian physicist, and later in 1923 by Pierre Auger, a French physicist.
What is the difference between Auger electron and secondary electron?
Auger Electrons have a characteristic energy, unique to each element from which it was emitted from. … Since a lower (usually K-shell) electron was emitted from the atom during the secondary electron process an inner (lower energy) shell now has a vacancy.
Which energy is responsible to release electrons in XPS?
In XPS, the sample is irradiated with low-energy (~1.5 keV) X-rays, in order to provoke the photoelectric effect. The energy spectrum of the emitted photoelectrons is determined by means of a high-resolution electron spectrometer.
Is XPS quantitative?
Like all other measurement methods, XPS is quantitative if the instrument is calibrated and reference materials or data are used.
What is the maximum depth of XPS?
XPS / ESCA The average depth of analysis for an XPS measurement is approximately 5 nm. PHI XPS instruments provide the ability to obtain spectra with a lateral spatial resolution as small as 7.5 µm.
How much sample do you need for XPS?
About 5 – 10 mg is the maximum amount that will be used even for large spot XPS analysis. Powder samples are not good for XPS analysis. If you make a pellet out of the powder and sinter it, it will work better.
What is the difference between XRD and EDX?
EDX is used for chemical identification of elements and their concentration. X-Ray Diffraction (XRD) is used for Phase identification. It is used to identify spatial arrangements of atoms in crystalline phases. In EDX, electrons knock out electrons from atoms, producing X-rays of characteristic wavelength.
Is XPS a bulk technique?
Given that surface Si concentrations as low as 0.10. % can be detected, the advantage of XPS over bulk techniques is readily apparent. One very important reason for using XPS Surface Aanlysis is that it is nondestructive.
Is XRD surface sensitive?
XRD examines the crystallinity of a sample. … X-ray photoelectron spectroscopy (XPS) is a surface sensitive technique or normally called ESCA ( (Electron Spectroscopy for Chemical Analysis) ), used to study the electronic states and chemical composition of the surface of a sample.
What is valence Spectroscopy?
Valence band spectra (VBS) can be calculated from first principles for comparison. … They give information about the density and occupancy of electronic states in the valence band of the material.
How is spectroscopy used in chemical analysis?
Spectroscopy is used as a tool for studying the structures of atoms and molecules. … In a typical spectroscopic analysis, a concentration of a few parts per million of a trace element in a material can be detected through its emission spectrum.
What is ESCA and application?
Electron Spectroscopy for Chemical Analysis (ESCA), also called X-ray Photoelectron. Spectroscopy (XPS), is a surface analysis technique that provides composition and chemical. bonding information on the surface of the sample. ESCA can detect all elements except. hydrogen and helium.