What is needed for electroplating?
.
Beside this, what is the procedure of electroplating?
Electroplating involves passing an electric current through a solution called an electrolyte. This is done by dipping two terminals called electrodes into the electrolyte and connecting them into a circuit with a battery or other power supply.
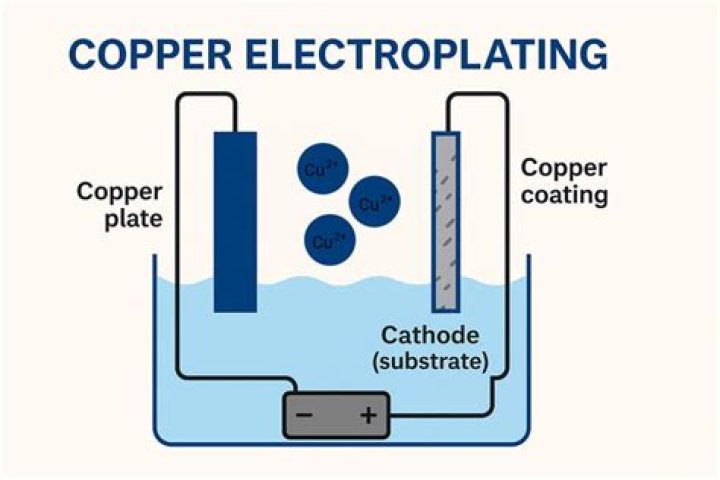
Furthermore, what is electroplating with example? A simple example of the electroplating process is the electroplating of copper in which the metal to be plated (copper) is used as the anode, and the electrolyte solution contains the ion of the metal to be plated (Cu2+ in this example). Copper goes into solution at the anode as it is plated at the cathode.
In respect to this, why do we need electroplating?
Electroplating is used to apply metal coating on non metallic surfaces through electro chemical processes. Electroplating forms a protective barrier, reduces friction, and prevents tarnishing to name some of its benefits. Besides protecting metals from corrosion, electroplating can be used for: Decorative purposes.
What are the types of electroplating?
The different types of electroplating are: mass plating (also barrel plating), rack plating, continuous plating, and in line plating.
Related Question AnswersWhat are the disadvantages of electroplating?
There are a number of disadvantages of this process as well, such as: Non-uniform plating: electroplating may or may not be uniform and this may result in a substandard appearance of the plated material. Cost: the process is costly and time consuming.Can I electroplate at home?
To electroplate with this method, you will need a piece of copper, the metal to be plated, vinegar, hydrogen peroxide, alligator clips, a 6-volt lantern battery, a glass/plastic container, and gloves. All of these items can be purchased at a local hardware or home improvement store.What factors affect electroplating?
There are many factors that affect this process. The surface area of the electrodes, the temperature, the kind of metal and the electrolyte, the magnitude of the applied current are some of these factors. In this essay the factors that affect the electroplating process will be investigated.What are the advantages of electroplating?
There are a number of advantages of this process: Corrosion resistance: a corrosion-prone substance such as iron can be coated with a layer of non-corrosive material, thereby protecting the original material. Decorative items: shine and luster can be imparted to otherwise dull surfaces.How thick can you electroplate?
The maximum thickness of electroless nickel plating is limited to approximately 0.1 mm. Electroplating is the only possibile way to achieve a greater degree of thickness, which we define as "thick nickeling" or "thick nickel plating".What is electroplating with diagram?
ANSWER : The process in which the cations of metal are dissolved to form a thin coat over an electrode with the help of passing electric current is known as electroplating. The main use of electroplating method is to change the properties of any object like making the object resistant to corrosion.What is electrodeposition method?
Electrodeposition is a well-known method to produce in situ metallic coatings by the action of an electric current on a conductive material immersed in a solution containing a salt of the metal to be deposited.How electroplating is used in daily life?
Electroplating is the process of coating one metal with another metal to enhance its look. Articles used in daily life where electro plating is used: Nickel coating is done in articles like decorative items, cars and machinery parts.What are some applications of electroplating?
Applications of electroplating- Increase wear resistance.
- Protect against surface abrasions.
- Reduce friction.
- Improve electrical conductivity (copper layer onto an electrical component)
- Prepare surfaces for better adhesion before painting or re-coating.
Where is electroplating used?
Electroplating is used in jewelry making to coat base metals with precious metals to make them more attractive and valuable and sometimes more durable. Chromium plating is done on vehicle wheel rims, gas burners, and bath fixtures to confer corrosion resistance, enhancing the life expectancy of the parts.What industries use electroplating?
Modern electroplating is a form of metal finishing used in various industries, including aerospace, automotive, military, medical, RF microwave, space, electronics and battery manufacturing. It is the electrochemical process whereby metal ions in solution are bonded to a metal substrate via electrodeposition.What is the conclusion of electroplating?
conclusion on electroplating. Dear Student , Electroplating gives a metal protection from corrosion , as it is a process of coating the base metal with another metal . The base metal is inexpensive but the another metal used here is pure metal or one of several alloys .What are two uses of electroplating?
What is electroplating? List four uses of electroplating. Giving the reason- Tin cans used for storing food are made by electroplating tin metal on to iron.
- it is used in bicycle to resist rust or corrosion.
- it is used in jewellery to make it lusturous.
- it is used in LPG stoves to resist rust.