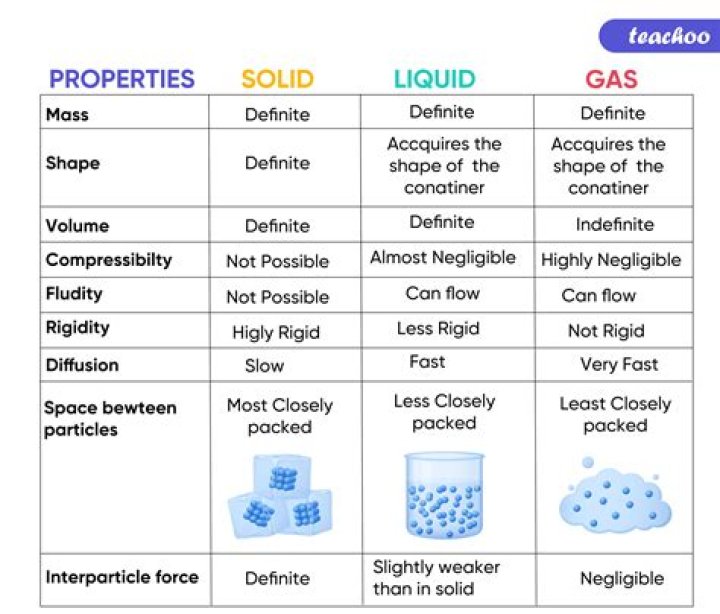
The process of conversion of gas directly to solid state is called Deposition . It is the reverse of Sublimation , which is transition of a substance directly from solid to gaseous phase..
Simply so, what is an example of deposition gas to solid?
Deposition refers to the process in which a gas changes directly to a solid without going through the liquid state. For example, when warm moist air inside a house comes into contact with a freezing cold windowpane, water vapor in the air changes to tiny ice crystals.
Also Know, what is the process of conversion of solid state directly to gaseous state called? The process in which a solid directly changes into gaseous state is called evaporation.
People also ask, can gas be converted into solid?
Under certain circumstances, gas can transform directly into a solid. This process is called deposition. Water vapor to ice - Water vapor transforms directly into ice without becoming a liquid, a process that often occurs on windows during the winter months.
What is sublimation give three examples?
Sublimation is a type of phase transition, or a change in a state of matter, just like melting, freezing, and evaporation. Through sublimation, a substance changes from a solid to a gas without ever passing through a liquid phase. Dry ice, solid CO2, provides a common example of sublimation.
Related Question Answers
What are 3 examples of sublimation?
To help you gain a better understanding of this process, here are some real-life examples of sublimation: - Dry Ice. As mentioned earlier, dry ice is one of the most popular examples of sublimation in real life.
- Water.
- Specialized Printers.
- Moth Balls.
- Freeze Drying.
- Air Fresheners.
How is sublimation possible?
Through sublimation, a substance changes from a solid to a gas without ever passing through a liquid phase. Dry ice, solid CO2, provides a common example of sublimation. It is also possible for ice to sublimate, though it requires specific weather environments and high altitudes.What phase change is sublimation?
Sublimation Definition. Sublimation is the transition from the solid phase to the gas phase without passing through an intermediate liquid phase. This endothermic phase transition occurs at temperatures and pressures below the triple point.What is a real life example of sublimation?
There are many examples of sublimation in daily life: The air fresheners used in toilets. The solid slowly sublimes and releases the pleasant smell in the toilet over a certain period of time. Moth balls, made of naphthalene are used to drive away moths and some other insects.What is sublimation in states of matter?
Sublimation is a type of phase transition, or a change in a state of matter, just like melting, freezing, and evaporation. Through sublimation, a substance changes from a solid to a gas without ever passing through a liquid phase. Dry ice, solid CO2, provides a common example of sublimation.Is dry ice sublimation or deposition?
The opposite process is called deposition, where CO2 changes from the gas to solid phase (dry ice). At atmospheric pressure, sublimation/deposition occurs at −78.5 °C (−109.3 °F) or 194.65 K. The density of dry ice varies, but usually ranges between about 1.4 and 1.6 g/cm3 (87 and 100 lb/cu ft).What is the opposite of sublimation?
Sublimation is the transition from the solid to the gas state. A good example is carbon dioxide ice. The opposite of this process is called deposition : the transition from the gas to the solid. Condensation is the transition from gas to liquid as in the condensation of steam to liquid water.What is meant by melting?
Melting is the process by which a substance changes from the solid phase to the liquid phase. Melting occurs when the internal energy of a solid increases, usually through the application of heat or pressure, such that the molecules become less ordered.What is a gas to a liquid called?
Condensation- It is the change of the physical state of matter from gas phase into liquid phase. Evaporation is a type of vaporization of a liquid that occurs from the surface of a liquid into a gaseous phase that is not saturated with the evaporating substance.Is sublimation endothermic or exothermic?
Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and deposition are exothermic processes. Changes of state are examples of phase changes, or phase transitions. All phase changes are accompanied by changes in the energy of a system.What is sublimation and deposition?
Sublimation and deposition are opposite processes. Sublimation is when a substance goes from solid to gas while deposition is when a substance goes from gas to solid. Sublimation and deposition chemistry are examples of phase changes. This is evaporation, or the liquid phase of water going to the vapor phase of water.What is phase change diagram?
Phase diagram is a graphical representation of the physical states of a substance under different conditions of temperature and pressure. As we cross the lines or curves on the phase diagram, a phase change occurs. In addition, two states of the substance coexist in equilibrium on the lines or curves.Can a gas turn into a liquid?
Technically, a gas can not be turned into a liquid. This is part of the definition of a gas. A gas is above its critical temperature, by definition. Nothing above its critical temperature can be reduced to a liquid by compression.What is it called when a solid turns into a liquid?
The solid begins to go from a solid state to a liquid state — a process called melting. The temperature at which melting occurs is the melting point (mp) of the substance. The melting point for ice is 32° Fahrenheit, or 0° Celsius.What is it called when gas turns into plasma?
Ionization (Gas → Plasma) This image captures the ionization of particles in the upper atmosphere to form the aurora. Ionization may be observed inside a plasma ball novelty toy. Ionization energy is the energy required to remove an electron from a gaseous atom or ion.What is an example of a solid to gas?
Some examples of this process are the following: Dry ice is a solid made from carbon dioxide gas undergoes sublimation thus there is no remnants of liquid form. There is no more need to drain the container of its liquid contents. Solid car air fresheners hanged inside cars change directly into vapor without melting.How is sublimation different from evaporation?
Although both these processes describe the conversion of matter into the gaseous phase, the terms are different from each other. The main difference between sublimation and evaporation is that sublimation is the phase transition from solid to gas whereas evaporation is the phase transition from liquid to gas.What is an example of a solid?
Other solids, known as amorphous solids, lack any apparent crystalline structure. Examples of solids are common table salt, table sugar, water ice, frozen carbon dioxide (dry ice), glass, rock, most metals, and wood. Water ice is an example of a solid that becomes liquid when it is heated gradually.