What is concept of entropy?
.
Similarly, what is a simple definition of entropy?
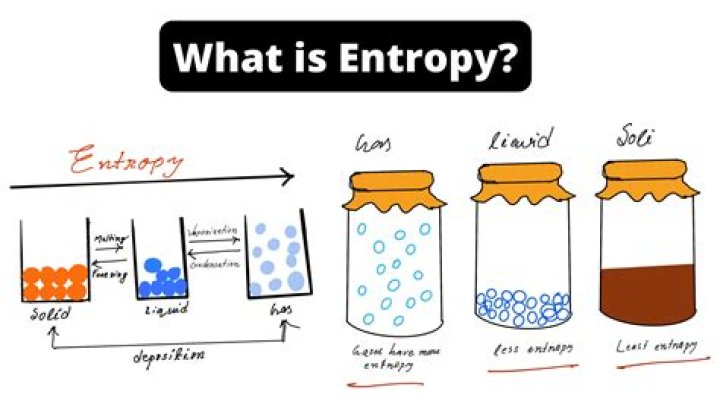
The entropy of an object is a measure of the amount of energy which is unavailable to do work. Entropy is also a measure of the number of possible arrangements the atoms in a system can have. In this sense, entropy is a measure of uncertainty or randomness.
Secondly, what is the concept of enthalpy? Enthalpy is a thermodynamic property of a system. It is the sum of the internal energy added to the product of the pressure and volume of the system. However, it is possible to measure the difference in enthalpy between one state and another. Enthalpy change may be calculated under conditions of constant pressure.
In this regard, what is entropy with example?
A campfire is an example of entropy. The solid wood burns and becomes ash, smoke and gases, all of which spread energy outwards more easily than the solid fuel. Ice melting, salt or sugar dissolving, making popcorn and boiling water for tea are processes with increasing entropy in your kitchen.
What is entropy and its unit?
Entropy is the measure of the disorder of a system. It is an extensive property of a thermodynamic system, which means its value changes depending on the amount of matter that is present. In equations, entropy is usually denoted by the letter S and has units of joules per kelvin (J⋅K−1) or kg⋅m2⋅s−2⋅K−1.
Related Question AnswersWhat is the opposite of entropy?
Negentropy is reverse entropy. It means things becoming more in order. By 'order' is meant organisation, structure and function: the opposite of randomness or chaos. One example of negentropy is a star system such as the Solar System. The opposite of entropy is negentropy.What is the first law of entropy?
The first law, also known as Law of Conservation of Energy, states that energy cannot be created or destroyed in an isolated system. The second law of thermodynamics states that the entropy of any isolated system always increases.What is another word for entropy?
Synonyms for entropy | noundeterioration breakup. collapse. decay. decline. degeneration.What causes entropy?
Several factors affect the amount of entropy in a system. If you increase temperature, you increase entropy. (1) More energy put into a system excites the molecules and the amount of random activity. (2) As a gas expands in a system, entropy increases.Why is entropy so important?
Entropy is a bookkeeping device, which tells us about the flow and distribution of energy. For any process to occur to occur spontaneously, it is a necessary condition that the entropy of the system undergoing the process should increase. If the entropy decreases, then that process cannot occur spontaneously.How does entropy explain life?
"Living organisms preserve their internal order by taking from their surroundings free energy, in the form of nutrients or sunlight, and returning to their surroundings an equal amount of energy as heat and entropy."What are the three laws of thermodynamics?
The three laws of thermodynamics define physical quantities (temperature, energy, and entropy) that characterize thermodynamic systems at thermodynamic equilibrium. The laws describe how these quantities behave under various circumstances, and preclude the possibility of certain phenomena (such as perpetual motion).What is entropy vs enthalpy?
Scientists use the word entropy to describe the amount of freedom or randomness in a system. In other words, entropy is a measure of the amount of disorder or chaos in a system. Entropy is thus a measure of the random activity in a system, whereas enthalpy is a measure of the overall amount of energy in the system.Is entropy a force?
In physics, an entropic force acting in a system is an emergent phenomenon resulting from the entire system's statistical tendency to increase its entropy, rather than from a particular underlying force on the atomic scale. It should not be referred to as a distribution of the entropy in the space.Can entropy be negative?
Entropy is the amount of disorder in a system. Negative entropy means that something is becoming less disordered. In order for something to become less disordered, energy must be used. The second law of thermodynamics states that the world as a whole is always in a state of positive entropy.How is enthalpy used in real life?
Refrigerator compressors and chemical hand warmers are both real-life examples of enthalpy. Both the vaporization of refrigerants in the compressor and the reaction to the iron oxidation in a hand warmer generate a change in heat content under constant pressure.What is enthalpy with example?
Enthalpy and chemical reactions For example, if gasoline is burned in the open air, heat is released by the gasoline. If we suppose 100 kilojoules of heat were released, then the enthalpy of the gasoline was reduced by 100 kilo joules. Therefore the change in enthalpy for this reaction was ∆H = –100 kJ.Who invented entropy?
Rudolf ClausiusWhat is the equation for entropy?
If the process is reversible, then the change in entropy is equal to the heat absorbed divided by the temperature of the reversible process. In the equation, Q is the heat absorbed, T is the temperature, and S is the entropy. Entropy is also the measure of energy not available to do work for your system.How do you use Entropy in a sentence?
Entropy in a Sentence ??- Sue prevents her small apartment from falling into entropy by storing items in containers and on shelves.
- With the teacher in the hallway, the classroom descended into entropy.
- The older Ted became, the faster his body fell into entropy.