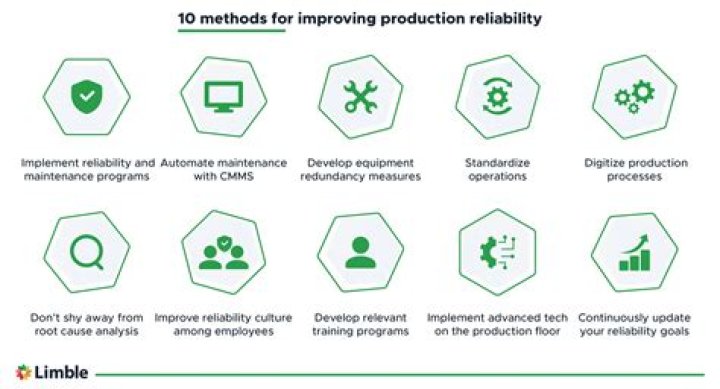
What is acids bases and salts in chemistry
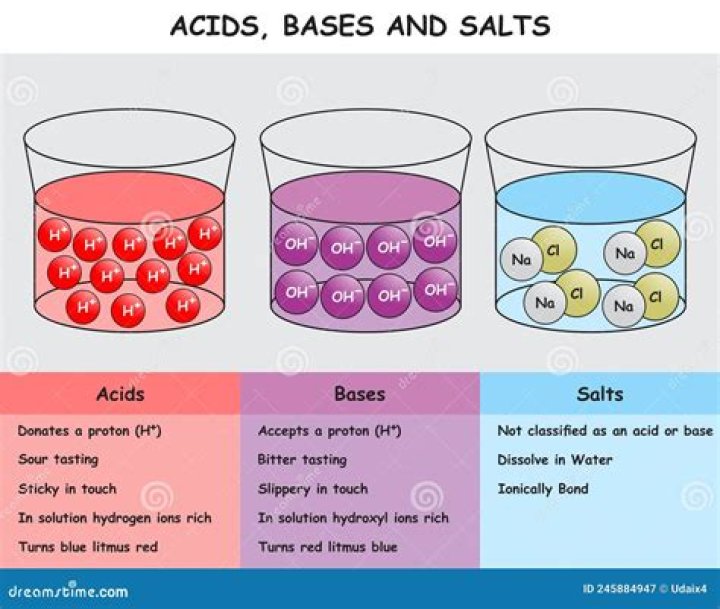
An acid is defined as a substance whose water solution tastes sour, turns blue litmus red and neutralizes bases. … Salt is a neutral substance whose aqueous solution does not affect litmus. According to Faraday: acids, bases, and salts are termed as electrolytes.
What is difference between acid base salt?
ACIDSBASESAcid are those that can donate electronsBases are those that can accept protonsThe pH values of acids are basically less than 7The pH values of bases are basically more than 7.Acids turn blue litmus red.The Base turns red litmus blue.
What is an base in chemistry?
base, in chemistry, any substance that in water solution is slippery to the touch, tastes bitter, changes the colour of indicators (e.g., turns red litmus paper blue), reacts with acids to form salts, and promotes certain chemical reactions (base catalysis).
What are acids bases and salts for Class 7?
Section NameTopic Name5Acids, Bases and Salts5.1Acids and Bases5.2Natural Indicators Around us5.3NeutralisationWhat is called base?
A base is a substance that can neutralize the acid by reacting with hydrogen ions. Most bases are minerals that react with acids to form water and salts. Bases include the oxides, hydroxides and carbonates of metals. The soluble bases are called alkalis. … Therefore, All alkalis are bases, but not all bases are alkalis.
What are salts in chemistry?
salt, in chemistry, substance produced by the reaction of an acid with a base. A salt consists of the positive ion (cation) of a base and the negative ion (anion) of an acid. … The term salt is also used to refer specifically to common table salt, or sodium chloride.
What is difference between acids and bases?
Acid is a kind of chemical compound that when dissolved in water gives a solution with H+ ion activity more than purified water. A base is an aqueous substance that donates electrons, accept protons or release hydroxide (OH-) ions.
Is pH an acid or base?
pH is a measure of how acidic/basic water is. The range goes from 0 – 14, with 7 being neutral. pHs of less than 7 indicate acidity, whereas a pH of greater than 7 indicates a base.What does pH stand for?
pH may look like it belongs on the periodic table of elements, but it’s actually a unit of measurement. The abbreviation pH stands for potential hydrogen, and it tells us how much hydrogen is in liquids—and how active the hydrogen ion is.
What is the difference between acids and bases Class 7th?The difference between the acids and bases are: Acids are sour in taste and bases are bitter in taste. Acids turn blue litmus to red, but bases do not change the colour of blue litmus.
Article first time published onWhat is an acid Class 10?
Acids: Acids are sour in taste, turn blue litmus red, and dissolve in water to release H+ ions. Example: Sulphuric acid (H2SO4), Acetic Acid (CH3COOH), Nitric Acid (HNO3) etc. … Acids have a sour taste. Turns blue litmus red. Acid solution conducts electricity.
What is class 7th base?
The substances that taste bitter are called base and their nature is basic. They are slippery to touch. Example: baking soda containing sodium hydrogen carbonate, Milk of magnesia, Soaps, Lime water containing calcium hydroxide, Household cleaners.
Why are bases called caustic?
Explanation: Usually these bases are created by adding pure alkali metals such as sodium into the conjugate acid. They are called superbases, and it is impossible to keep them in water solution because they are stronger bases than the hydroxide ion.
What are bases used for?
Some Common BasesUsessodium hydroxide, NaOH (lye or caustic soda)Used in the manufacture of soaps and detergents, and as the main ingredient in oven and drain cleaners.potassium hydroxide, KOH (lye or caustic potash)Used in the production of liquid soaps and soft soaps. Used in alkaline batteries.
How are bases named?
How are bases named? Bases are named in the same way as other ionic compounds-the name of the cation is followed by the name of the anion.
What are the two types of bases?
Bases can primarily be divided into two types: Strong bases and Weak bases.
What are three differences between acids and bases?
Bases usually exist in the solid-state except for ammonia which exists as a gas. Acids are sour in taste. Bases taste bitter. Acids release hydrogen ions (H+) when dissolved in water.
Is milk an acid or base?
Cow’s milk Milk — pasteurized, canned, or dry — is an acid-forming food. Its pH level is below neutral at about 6.7 to 6.9. This is because it contains lactic acid. Remember, though, that the exact pH level is less important than whether it’s acid-forming or alkaline-forming.
What is acid and base explain with example?
(a) Acids are those chemical substances that have a sour taste. Example: Acetic acid and citric acid. Base is a chemical substance that has a bitter taste. Example: Caustic soda and washing soda. (b) Strong bases – Sodium hydroxide (NaOH), potassium hydroxide (KOH).
What is salt Short answer?
In chemistry, a salt is any neutral chemical compound made of cations (positive ions) attached to anions (negative ions). The main kind of salt is sodium chloride with the chemical formula NaCl. It is formed when hydrochloric acid is added in sodium hydroxide.
What is salt and its types?
The different types of salts are: normal salt, acid salt, basic salt and double salt. 1. Normal salt : A salt that does not contain any replaceable hydrogen atoms or hydroxyl groups is called normal salt.
What are the examples of basic salts?
- Calcium carbonate (CaCO3)
- Sodium acetate (NaOOCCH3)
- Potassium cyanide (KCN)
- Sodium sulfide (Na2S)
What is the pH of blood?
The acidity and alkalinity of your blood are measured using the pH scale. The pH scale ranges from 0 (very acidic) to 14 (very alkaline). Blood is usually between 7.35 to 7.45.
What is a pH of 0?
A solution that has a pH of 0 is very acidic with a high concentration of hydrogen ions (although things can be of greater acidity), a pH of 7 is a neutral solution, equal concentrations of hydrogen ions and their basic counterpart hydroxy ions. … The H in pH stands for hydrogen.
What is pH level in body?
The pH scale, ranges from 0 (strongly acidic) to 14 (strongly basic or alkaline). A pH of 7.0, in the middle of this scale, is neutral. Blood is normally slightly basic, with a normal pH range of about 7.35 to 7.45. Usually the body maintains the pH of blood close to 7.40.
Is lemon juice basic or acidic?
Lemon juice has a pH between 2 and 3, which means it’s 10,000–100,000 times more acidic than water (1, 2, 3). A food’s pH is a measure of its acidity. The pH of lemon juice falls between 2 and 3, meaning it is acidic.
Is ammonia a base?
Ammonia is a typical weak base. Ammonia itself obviously doesn’t contain hydroxide ions, but it reacts with water to produce ammonium ions and hydroxide ions. … A weak base is one which doesn’t convert fully into hydroxide ions in solution.
How do acids work?
An acid dissociates, or breaks apart, and donates protons, or hydrogen ions, in an aqueous solution, while a base donates hydroxide ions in a solution. … When acids are added, they release more hydrogen ions into the solution, and this causes the pH of the solution to drop.
What are the differences between acids and bases Brainly?
ACIDS HAVE SOUR IN TASTE WHEREAS BASE HAVE BITTER IN TASTE ACIDS PH VALUE IS LESS THAN 7 BUT BASE HAVE THE PH VALUE IS MORE THAN 7 ACIDS TURNS BLUE LITIMUS TO RED WHEREAS BASE TURNS RED TO BLUE. HCL, HNO3, CH3COOH, ETC ARE SOME EXAMPLES OF ACIDS AND NAOH, MG(OH) 2, ETC ARE SOME EXAMPLES OF BASE.
Is distilled water acidic?
Solution 4: Distilled water is neutral.
What is acid and base class 8?
Definition. An acid is any chemical compound once dissolved in water produces a solution with hydrogen ion activity more than purified water. A base is an aqueous substance that could absorb hydrogen ions. Strength.