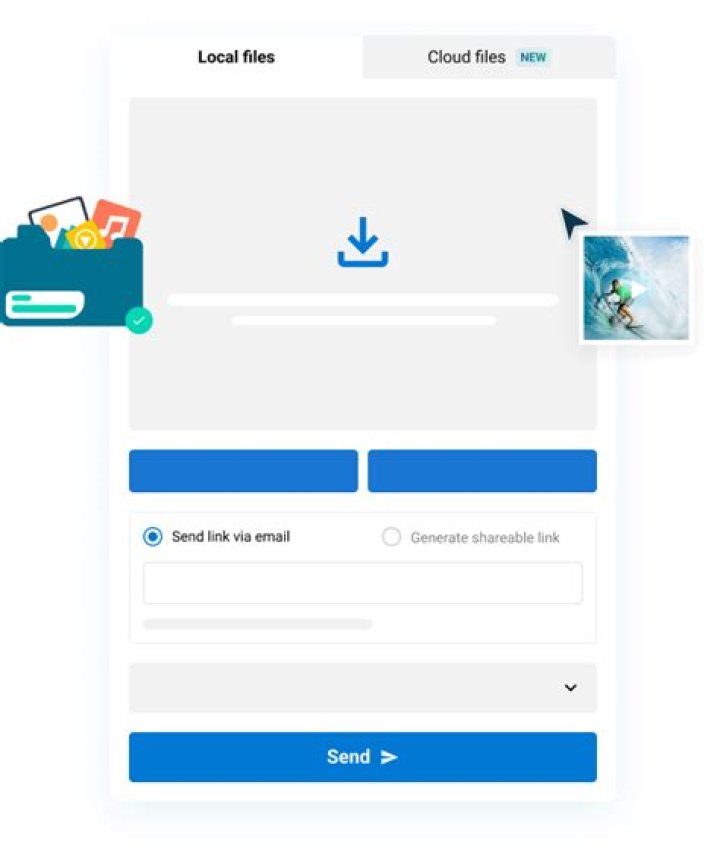
In SI base units:s−1.
Subsequently, one may also ask, what is the activity of a substance?
In chemical thermodynamics, activity (symbol a)is a measure of the “effective concentration” of aspecies in a mixture, in the sense that the species' chemicalpotential depends on the activity of a real solution in thesame way that it would depend on concentration for an idealsolution.
Subsequently, question is, what is activity and activity coefficient? Published on Nov 7, 2017. ACTIVITY IS THEEFFECTIVE CONCENTRATION OF THE SPECIES PRESENT IN THE SOLUTION ANDACTIVITY COEFFICIENT IS THE RATIO OF ACTIVITY ANDCONCENTRATION.
In this way, what is the units of equilibrium constant?
Therefore the unit for the equilibriumconstant is that of a concentration, in SI that would bemolm−3. It is derived from the Law of mass action; thederivation can be found on Wikipedia and various othersources.
Why is K Unitless?
Since activities are unitless, they eliminate theunits of all the quantities in the equilibrium constant expression,making the constant itself unitless all the time. Theactivity of a substance is a measure of the degree of presence of achemical, relative to a reference state.
Related Question Answers
Can activity coefficient be less than 1?
The activity coefficient γican be greater than one or less than one andis a function of composition, temperature, and pressure. Equation(11-157) shows that activity coefficients less than unityare due to negative deviations from Raoult's law, that is, theactivity of a component is smaller than its molefraction.What is activity concentration?
In chemical thermodynamics, activity (symbol a)is a measure of the "effective concentration" of a speciesin a mixture, in the sense that the species' chemical potentialdepends on the activity of a real solution in the same waythat it would depend on concentration for an idealsolution.What are the units of K?
The units of k depend on the order of thereaction, but the units are never Newtons per metre. As anexample, for a first order reaction, k has the unitsof 1/s and for a second order reaction, units of1/M.s.What is the formula of equilibrium constant?
The value of R is .08206 L atmmol-1 K-1. T representsthe temperature in Kelvin (K) at which the equilibriumreaction takes place. To find Δngas , simply subtract the sumof the coefficients of the reactants from the sum of thecoefficients of the products. The value Δngas is (1+2)-1 =2.Does constant K have units?
What is the unit of the specific rateconstant k? It depends on the order of the reaction. kunits: M/s, M/min, M/hr, etc. k units: s-1, min-1, hr-1,etc.What does KEQ mean?
Illustrated Glossary of Organic Chemistry - Equilibriumconstant (Keq) Equilibrium constant (Keq): Aratio that quantifies the position of a chemical equilibrium. Whenthe equilibrium favors reactants ΔG > 0 and Keq< 1.What is the importance of equilibrium constant?
Equilibrium constants can be vitallyimportant in the context of chemical reactions as it tellsyou how reversible a reaction is. The equilibrium constant,K, is the concentration of the reactants over the concentration ofproducts once the reaction has reachedequilibrium.What is the difference between K and Q?
The difference between K and Q is that, Kis the constant of a certain reaction when it is in equilibrium,while Q is the quotient of activities of products andreactants at any stage of a reaction. Therefore, by comparingQ and K, we can determine the direction of areaction.What is K in chemistry?
1.K IS THE CHEMICAL SYMBOL OF ELEMENTPOTASSIUM. 2.K INDICATES OR CAN BE CALLED THAT K ISTHE SI UNIT OF TEMPRETURE (0 DEGREE CENTIGRATE =273 KELVIN. MOST OFTEMPRETURES IN CHEMISTRY ARE EXPRESSED IN KELVIN. 3.KIS EQUILIBRIUM CONSTANT. 4.IN ANY CHEMICAL REACTION FREEENERGY IS EXPRESSED IN K.Cals WHERE K MEANSKILO.What does rate constant mean?
The rate constant, or the specific rateconstant, is the proportionality constant in theequation that expresses the relationship between the rate ofa chemical reaction and the concentrations of the reactingsubstances.What affects equilibrium constant?
Equilibrium constants are not changed if you add(or change) a catalyst. The only thing that changes anequilibrium constant is a change of temperature. Theposition of equilibrium is not changed if you add (orchange) a catalyst. A catalyst speeds up both the forward and backreactions by exactly the same amount.What is an activity measure?
Activity Measure Definition: An allocation basein an activity-based costing system; ideally, ameasure of the amount of activity that drives thecosts in an activity cost pool.What is an activity in Android?
Last update: 2014-10-30. An Android activity isone screen of the Android app's user interface. In that wayan Android activity is very similar to windows in a desktopapplication. An Android app may contain one or moreactivities, meaning one or more screens.