Is silver bromide A compound or element?
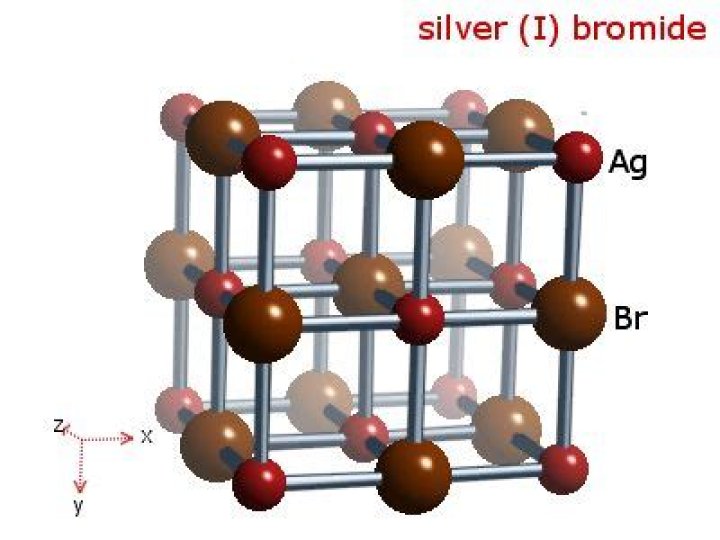
.
Moreover, what is the chemical formula for silver bromide?
AgBr
One may also ask, what is bromide made of? A bromide is a chemical compound containing a bromide ion or ligand. This is a bromine atom with an ionic charge of −1 (Br−); for example, in caesium bromide, caesium cations (Cs+) are electrically attracted to bromide anions (Br−) to form the electrically neutral ionic compound CsBr.
Just so, what happens to silver bromide in light?
When silver bromide or AgBr is kept in sunlight a chemical reaction occurs. Chemical decomposition called photo chemical reaction occurs where silver bromide turns into grey colour. When we keep the silver bromide in the sunlight then after 2 to 3 Hours it became in Grey colour.
Why is silver bromide used in photographic film?
Due to this property silver salts are used in photography. When silver bromide is exposed to light it undergoes photochemical decomposition. The silver ions present in silver bromide are reduced to silver metal which remains as an opaque image on the photographic film.
Related Question Answers