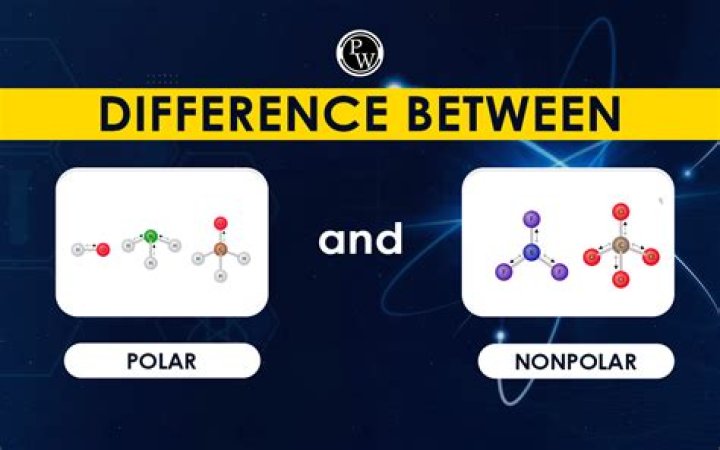
Is salad dressing polar or nonpolar?
.
Accordingly, is vinegar polar or nonpolar?
Vinegar is composed of acetic acid and water, which are polar compounds. In a polar molecule, one or a group of atoms have a stronger pull on the electrons in the molecule.
Subsequently, question is, why does my salad dressing separate? No matter how hard you try to shake, stir, or whisk oil and vinegar together, they eventually separate. This happens because vinegar and oil are made of very different types of molecules that are attracted to their own kind.
Also to know is, is salad dressing a compound?
Answer and Explanation: Salad dressing can be a homogeneous mixture or a heterogeneous mixture based on the type of salad dressing it is.
Which flavor ingredients will not leave sediments in the salad dressing?
Lecithin because it did not leave any sediment and it did not have any layers so that means it dissolved and will make a better salad dressing.
Related Question AnswersIs water Polar?
A water molecule, because of its shape, is a polar molecule. That is, it has one side that is positively charged and one side that is negatively charged. The molecule is made up of two hydrogen atoms and one oxygen atom. The bonds between the atoms are called covalent bonds, because the atoms share electrons.Is NaCl a polar molecule?
Yes, NaCl is an ionic bond which makes it polar. A polar molecule consists of atoms having either a positive or negative bond. In this case, Na has a +1 charge and Cl has a -1 charge, making the bond polar. The difference in electronegativities is what makes a bond polar or nonpolar.Is nail polish non polar?
Nail polish has a number of ingredients, one of which is resin, which can be very non-polar (think of tree sap, for example). This is why acetone can remove nail polish even though water can't.What vinegar does to oil?
Oil and vinegar do not mix or even if they are mixed they will quickly separate when given the opportunity. Some proteins such as eggs are emulsifiers that will cause oil and vinegar to mix.Why is ch3cooh polar?
Since it is a double bond, the electrons on the oxygen make acetic acid extremely polar unless there exist excessive dipole cancellations in the same framework, which usually doesn't happen. In simple terms, it is a polar molecule, which y means that there is one positive and one negative end to the same.Is Vinegar an emulsifier?
Emulsifiers are substances that facilitate the dispersion of one phase (as tiny droplets) into another. A simple vinaigrette does not contain emulsifiers so the smaller volume of vinegar ends up being dispersed as droplets in a much larger continuous phase of oil.Why is it necessary to shake a bottle of salad dressing?
A bottle of salad dressing must be shaken well before use. This is necessary because it is a mixture of vegetable oil, which is non-polar and vinegar, which is polar. These polar and non-polar molecules do not mix. The chemical properties of the compounds greatly influence their solubility in the other compounds.Is ch3cooh a vinegar?
CH3COOH is an accepted and common form of writing the structure of acetic acid (commonly known as vinegar when diluted in water).Is blood a compound?
Explanation: Blood is a complex mixture of compounds, including the plasma and red and white blood cells. Chemically, they are proteins, or hydrocarbons.Is chocolate chip cookie homogeneous or heterogeneous?
A chocolate chip cookie is homogeneous because you can pull out the separate parts of the cookie. is distilled water heterogeneous or homo or a pure substance? Distilled water is homogeneous because there are different substances in the water but you can't see or pull the particles out of the water.Is lemonade a homogeneous mixture?
Homogeneous Mixture – appears uniform and consistent throughout. Solutions and colloids are homogeneous mixtures. A lemonade drink is a homogeneous mixture, known as a solution. So is air.Is air a homogeneous mixture?
Characteristics of mixtures A mixture in which its constituents are not distributed uniformly is called heterogeneous mixture, such as sand in water. One example of a mixture is air. Air is a homogeneous mixture of the gaseous substances nitrogen, oxygen, and smaller amounts of other substances.How do you combine oil and vinegar?
Mixing Techniques- Place the oil and vinegar in a jar with a tight-fitting lid and shake the jar vigorously for 30 seconds.
- Whisk the two liquids in a bowl with a wire whisk.
- Blend the oil and vinegar using an immersion or countertop blender or a food processor.