Is KCl a basic salt?
.
Consequently, is NaNO2 a basic salt?
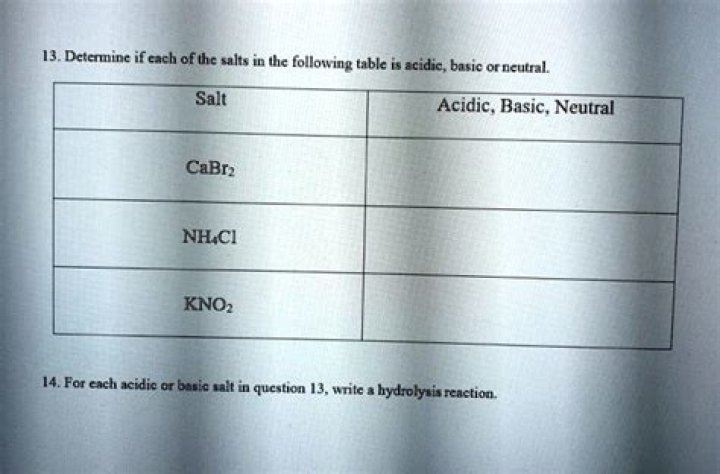
Classify each salt as acidic, basic, or neutral. K2SO4, NH4NO3, NaNO2. H2SO4 is a strong base and KOH is a strong acid. When a strong baseand a strong acid combine, they form a salt.
can we use KCl instead of NaCl? Nevertheless, in comparison with NaCl the saltiness of KCl is significantly lower when we compare the same mass concentrations. Despite all these objective disadvantages, KCl is still the best alternative for common salt (NaCl) substitution.
Accordingly, is k3po4 an acid or basic salt?
Classify these salts as acidic, basic, or neutral: NH4ClO4, K3PO4, Na2S, KCl, LiNO3. Acidic= Basic= Neutral=
What is the difference between potassium chloride and salt?
Both are salts. Here are some differences between Potassium chloride (KCl) and table salt: KCl has bitter taste; table salt has salty taste. KCl is more used as fertilizer; table salt is more used in food, as condiment or food preservative (food industry).
Related Question AnswersIs NaNO2 strong or weak?
K2SO4, NH4NO3, NaNO2. K2SO4 = neutral, it is a salt. H2SO4 is a strong base and KOH is a strong acid.What are strong bases?
Strong bases are able to completely dissociate in water- LiOH - lithium hydroxide.
- NaOH - sodium hydroxide.
- KOH - potassium hydroxide.
- RbOH - rubidium hydroxide.
- CsOH - cesium hydroxide.
- *Ca(OH)2 - calcium hydroxide.
- *Sr(OH)2 - strontium hydroxide.
- *Ba(OH)2 - barium hydroxide.
What kind of salt is KBr?
Potassium bromide, a typical ionic salt, is fully dissociated and near pH 7 in aqueous solution. It serves as a source of bromide ions. This reaction is important for the manufacture of silver bromide for photographic film: KBr(aq) + AgNO3(aq) → AgBr(s) + KNO3(aq)What type of salt is k2so4?
Potassium sulphate (chemical formula K2SO4) is prepared by the action of potassium hydroxide (KOH) and sulphuric acid (H2SO4). Since both the acid and the base forming the salt are strong, therefore potassium sulphate is a neutral salt.Is h3po4 a salt?
Salts: AgNO3(Salt), NaCl(Salt), NaNO3(salt), KNO3(salt), Acids: H3PO4(acid), HCl(acid), H2SO4(acid), HC2H3O2(acid), HNO3(acid), H2SO3(acid) Bases: Mg(OH)2(base), NaOH(base), KOHbase), NH4OH(base).What is a weaker base Cl or no2?
To find out which of the bases is weaker the only thing you need is to compare the atomic number of each element you got from the task. Then, the smaller number is obviously indicates the weaker base. So from the periodic table we know that atomic number of Cl is 17, and atomic number of No2 is sh.What is the Ka of no2?
| Ka | Acid | Base |
|---|---|---|
| 7.2 * 10-4 | Nitrous acid | Nitrite ion |
| 6.6 * 10-4 | Hydrofluoric acid | Fluoride ion |
| 1.8 * 10-4 | Methanoic acid | Methanoate ion |
| 6.3 * 10-5 | Benzoic acid | Benzoate ion |
Is NaCl an acid or base?
Expert Answers info NaCl is formed by the reaction of HCl and NaOH. Both are strong acids and bases. When a strong acid and a strong base react together the resultant is salt and water. Therefore NaCl is a salt.Is koh a weak base?
KOH is potassium hydroxide. Since it is composed of the hydroxide anion (OH-), it is a strong base. In solution, the hydroxide anion will completely react with any available protons, that is why KOH is a strong base. It is not an acid of any type, weak or strong, since KOH does not contribute any protons to solution.Is h3po4 a strong acid?
While phosphoric acid is quite acidic, it is evident that it is, indeed, a weak acid because of the lack of full dissociation in water; a 1 M solution of strong acid would be around 0 (0 for monoprotic species, possibly less for diprotic due to an additional hydrogen ion).Is NH4Cl a basic salt?
As mentioned in the other answer, NH4Cl is an “acidic” salt, formed by the neutralization of a strong acid (HCl) with a weak base (NH3). Therefore, when the salt is completely dissociated in an aqueous solution, it forms NH4+ and Cl- ions. Explanation: The solution of Ammonium Chloride is therefore acidic.Is NaF acidic or basic?
Similarly, NaF is basic (it is the salt of a strong base, NaOH, and a weak acid, HF). NaCl is neutral. This means that two of the solutions are basic (NH3 and NaF), one solution is neutral (NaCl), and the other is acidic (NH4Br).Is table salt acidic?
Properties of Table Salt: Table salt is the product formed by the neutralization of an acid by a base. So it is neither Acid nor Base. If the pH is less than 7, then it is acidic.What type of salt is k3po4?
Tripotassium phosphate, also called potassium phosphate tribasic, is a water-soluble salt which has the chemical formula K3PO4(H2O)x (x = 0, 3, 7, 9) Tripotassium phosphate is a strong base.Is HClO4 a strong acid?
HClO3 and HClO4 are strong acids due to the multiple oxygens bonded to the central atom. HClO2, however, is a weak acid.What are the examples of basic salt?
Other examples of basic salts include:- Calcium carbonate (CaCO3)
- Sodium acetate (NaOOCCH3)
- Potassium cyanide (KCN)
- Sodium sulfide (Na2S)