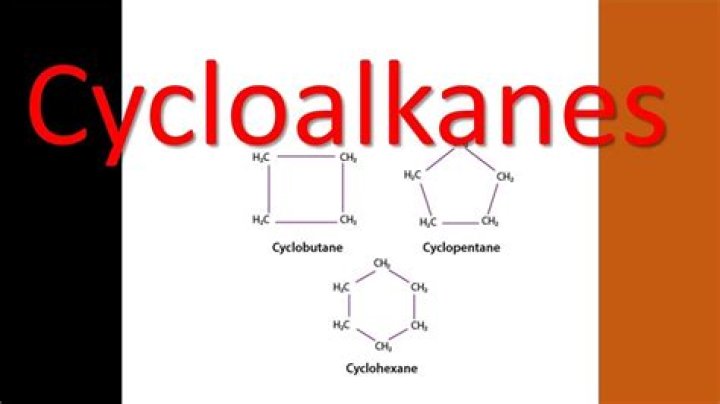
How do you name Cycloalkenes?
.
Simply so, what is the general formula for Cycloalkenes?
Cycloalkenes have the general formula CnH2(n-m). The letter m represents the number of double bonds. Thus, cyclopropene has the formula C3H4 while that of cyclobutene is C4H6. The properties of alkanes and alkenes are very similiar.
Also Know, how do you name Cycloalkanes? Naming Cycloalkanes Use the cycloalkane as the parent chain if it has a greater number of carbons than any alkyl substituent. b If an alkyl chain off the cycloalkane has a greater number of carbons, then use the alkyl chain as the parent and the cycloalkane as a cycloalkyl- substituent.
Considering this, how do you name an alkene ring?
Key Points
- Alkenes and alkynes are named by identifying the longest chain that contains the double or triple bond.
- The chain is numbered to minimize the numbers assigned to the double or triple bond.
- The suffix of the compound is “-ene” for an alkene or “-yne” for an alkyne.
Is benzene a Cycloalkene?
Benzene is an aromatic compound and Cycloalkene is an aliphatic cyclic compound. Yes benzene can be named as 1,3,5 cyclohexatriene . Aromatic compounds have different properties to that of alicyclic compounds hence we dont consider benzene as alicyclic system.
Related Question AnswersWhy are there no cyclic alkynes?
[Grade 12 Chemistry] Why are cyclic alkynes not often found and are often unstable? Cyclic alkynes do exist, but they are relatively more unstable than cyclic alkenes or alkanes because alkynes prefer a linear geometry.What is a cyclic alkene?
Acyclic alkenes, with only one double bond and no other functional groups, known as mono-enes, form a homologous series of hydrocarbons with the general formula CnH2n. Aromatic compounds are often drawn as cyclic alkenes, but their structure and properties are different and they are not considered to be alkenes.What is the other name of alkyne?
acetyleneIs pentane an alkane?
Pentane is an organic compound with the formula C5H12—that is, an alkane with five carbon atoms. Cyclopentane is not an isomer of pentane because it has only 10 hydrogen atoms where pentane has 12.What are the rules in naming alkenes?
The ene suffix (ending) indicates an alkene or cycloalkene. The longest chain chosen for the root name must include both carbon atoms of the double bond. The root chain must be numbered from the end nearest a double bond carbon atom.What is h3c?
CH3- is the symbol for methyl group. H3C is also a symbol for the same. It is sometimes written this way to show the direct bonding between the carbon and its neighbouring atom on the right hand. For eg: Methyl alcohol is written as CH3OH (which too is a correct representation of the compound).Is benzene an alkene?
Despite the presence of multiple double covalent bonds between Carbon atoms (C=C) in the Benzene molecule it is not considered an alkene, which by definition also contain such bonds.What is the formula for a cycloalkane?
Cycloalkanes with one ring have the general formula CnH2n compared with the general formula CnH(2n+2) for acyclic alkanes. Cycloalkanes have two fewer hydrogen atoms than alkanes, because another carbon-carbon bond is needed to form the ring.Which Cycloalkane is most stable?
It was pointed out by Sachse (1890) that cyclohexane exists in puckered forms - chair and boat forms - in these two puckered forms, the normal bond angle 109 deg 28 min is maintained throughout. Thus, among the most common cycloalkanes, cyclohexane is the most stable one.What is the simplest alkyne?
Alkynes are hydrocarbons containing carbon-carbon triple bond. They exhibit neither geometric nor optical isomerism. The simplest alkyne is ethyne (HCCH), commonly known as acetylene, as shown at right.What is the generic formula for an alkyne?
The most common alkyne is ethyne, better known as acetylene. The generic formula for alkanes is CnH2n+2, where n is the number identified by the prefix. Alkenes have the formula CnH2n and alkynes use the formula CnH2n-2.What is the simplest Cycloalkane?
The simplest cycloalkane is cyclopropane, a three-carbon ring. Figure 7.5.1: Cyclopropane is the simplest cycloalkane. Its highly strained geometry makes it rather unstable and highly reactive.What is the general formula of alkenes?
The alkenes comprise a series of compounds that are composed of carbon and hydrogen atoms with at least one double bond in the carbon chain. This group of compounds comprises a homologous series with a general molecular formula of C n H 2 n , where n equals any integer greater than one.Which compound is a Cycloalkene?
Alkenes are compounds that consist of carbons and hydrogens bonded together with a carbon-carbon double bond. Cycloalkenes are alkenes that consist of three or more carbon atoms linked together with at least one carbon-carbon double bond to form a structural ring (hence the prefix 'cyclo-') as shown below.What are the uses of Cycloalkanes?
Cycloalkanes can also be used for many different purposes. These uses are typically classified by the number of carbons in the cycloalkane ring. Many cycloalkanes are used in motor fuel, natural gas, petroleum gas, kerosene, diesel, and many other heavy oils.What is the general formula of a straight chain alkene with just one C C )?
Alkenes are unsaturated hydrocarbons containing at least one C=C bond. They have the general formula CnH2n, and are named similarly to alkanes (see Section 10.1.What are the first 10 alkenes?
The following is a list of the first ten Alkenes:- Ethene (C2H4)
- Propene (C3H6)
- Butene (C4H8)
- Pentene (C5H10)
- Hexene (C6H12)
- Heptene (C7H14)
- Octene (C8H16)
- Nonene (C9H18)