The reason that some inorganic ions have color is basically the same as for the organic molecules. Although no conjugated multiple bonds are involved, visible light is being absorbed and outer shell electrons are being promoted to higher energy levels, causing the perception of color..
Thereof, how do you tell if a system is conjugated?
If you see two double bonds separated by a single bond, their orbitals will interact and become conjugated!! o If the double bonds are separated by two sigma bonds, these atoms are not conjugated.
Furthermore, what is the difference between conjugation and Hyperconjugation? The two terms conjugation and hyperconjugation describe unsaturated organic compounds. The main difference between conjugation and hyperconjugation is that conjugation is the overlap of p-orbitals across a σ bond whereas hyperconjugation is the interaction of σ-bonds with a pi network.
In this way, what is meant by conjugated system?
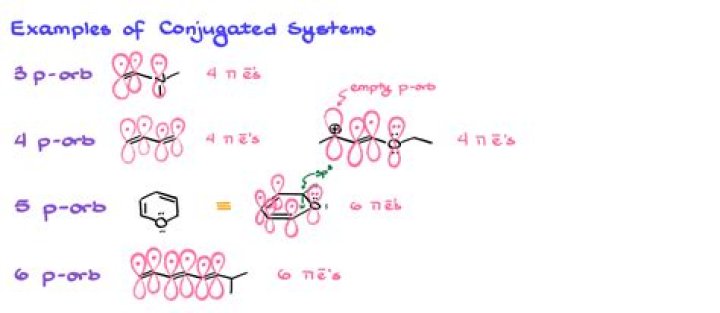
In chemistry, a conjugated system is a system of connected p orbitals with delocalized electrons in a molecule, which in general lowers the overall energy of the molecule and increases stability. It is conventionally represented as having alternating single and multiple bonds.
How many electrons are in a PI system?
The valence bond picture of 1,3-butadiene shows the two pi bonds as being isolated from one another, with each pair of pi electrons 'stuck' in its own pi bond. However, molecular orbital theory predicts (accurately) that the four pi electrons are to some extent delocalized, or 'spread out', over the whole pi system.
Related Question Answers
What is the conjugation?
Conjugation is the change that takes place in a verb to express tense, mood, person and so on. In English, verbs change as they are used, most notably with different people (you, I, we) and different time (now, later, before). Conjugating verbs essentially means altering them into different forms to provide context.What is resonance energy?
The resonance energy is defined as the difference between the electronic energy of a real (conjugated) molecule and a hypothetical Kekuléé structure with localized bonds.What is the difference between conjugation and resonance?
4. The Distinction Between Conjugation And Resonance. These conjugated pi systems contain electrons, which we often call “pi electrons” to distinguish them from the electrons that comprise single bonds in the molecule. The different arrangements of electrons within that “pi system” are called resonance forms.How do you determine hybridization?
A Shortcut For Determining The Hybridization Of An Atom In A Molecule - Look at the atom.
- Count the number of atoms connected to it (atoms – not bonds!)
- Count the number of lone pairs attached to it.
- Add these two numbers together.
What is pi electron?
Pi electron (π electron): An electron which resides in the pi bond(s) of a double bond or a triple bond, or in a conjugated p orbital. The allyl carbanion has four pi electrons.What are sigma and pi bonds?
Sigma and pi bonds are chemical covalent bonds. Sigma and pi bonds are formed by the overlap of atomic orbitals. A sigma bond, σ, resembles a similar "s" atomic orbital, and a pi pond, π, has the same orbital symmetry of the p orbital (again, in both cases when viewed down the bond axis).What is a delocalized bond?
Delocalization happens when electric charge is spread over more than one atom. For example, bonding electrons may be distributed among several atoms that are bonded together. Examples.How do you calculate electrons?
The number of electrons in a neutral atom is equal to the number of protons. The mass number of the atom (M) is equal to the sum of the number of protons and neutrons in the nucleus. The number of neutrons is equal to the difference between the mass number of the atom (M) and the atomic number (Z).Are lone pairs pi electrons?
The nitrogen on the bottom contributes it's lone pairs in resonance structures, therefore they do count as pi electrons. The total pi electron count = 6 (follows Huckel's Rule). This molecule is not aromatic. The lone pair cannot make a pi bond with the other carbons because it will create a pentacarbon (unstable).How many electrons are in a sigma bond?
1 Answer. anor277 · Truong-Son N. Well, formally, there are 6 electrons involved in the σ−bonding interaction .How many π electrons are there in benzofuran?
10 pi electrons
Are Annulenes aromatic?
Annulenes may be aromatic (benzene, [6]annulene and [18]annulene), non-aromatic ([8] and [10]annulene), or anti-aromatic (cyclobutadiene, [4]annulene). Thus, it does not exhibit appreciable aromaticity.How many pi electrons are in a triple bond?
When counting electrons to determine aromaticity, only two electrons are counted from the triple bond representing the π-bond that is conjugated with the ring, bringing the total count to 6 π-electrons.Are lone pairs sigma bonds?
Lone pairs of electrons are found in nonbonding orbitals, i.e. they are not being used to bond. Therefore, they do not count as σ -bonding pairs.What is Huckel rule of aromaticity?
In 1931, German chemist and physicist Erich Hückel proposed a theory to help determine if a planar ring molecule would have aromatic properties. His rule states that if a cyclic, planar molecule has 4n+2 π electrons, it is considered aromatic. This rule would come to be known as Hückel's Rule.What is Conjugative effect?
Conjugate effect (or delocalization) is an effect in which molecular orbitals (MOs) are conjugated to new molecular orbitals that are more delocalized and therefore generally lower in energy (the amount of MOs stays the same of course). The electrons can move freely in these new extended orbitals.What is drug conjugation?
ADC Review | Editorial Team. March 22, 2019. Antibody-drug Conjugates or ADCs are highly targeted biopharmaceutical drugs that combine monoclonal antibodies specific to surface antigens present on particular tumor cells with highly potent anti-cancer agents linked via a chemical linker.Why do conjugated systems absorb light?
For molecules having conjugated systems of electrons, the ground states and excited states of the electrons are closer in energy than for nonconjugated systems. This means that lower energy light is needed to excite electrons in conjugated systems, which means that lower energy light is absorbed by conjugated systems.Why are conjugated systems more stable?
Stability of Conjugated Dienes Conjugated dienes are more stable than non conjugated dienes (both isolated and cumulated) due to factors such as delocalization of charge through resonance and hybridization energy. Along with resonance, hybridization energy effect the stability of the compound.