How do bomb calorimetry and adiabatic calorimetry differ?
.
Keeping this in consideration, is a bomb calorimeter adiabatic?
A bomb calorimeter is only approximately adiabatic. In addition to measuring the energy release of one particular reaction, calorimetry is an important tool for determining the enthalpy of formation for the compound under study.
Furthermore, what is adiabatic calorimeter? Adiabatic calorimeters An adiabatic calorimeter is a calorimeter used to examine a runaway reaction. Since the calorimeter runs in an adiabatic environment, any heat generated by the material sample under test causes the sample to increase in temperature, thus fueling the reaction.
Also asked, what are the different types of calorimetry?
There are traditionally four types of reaction calorimeter- heat-flow, heat-balance, constant flux and power compensation calorimeters. These calorimeters can be used to measure both the exothermic and endothermic changes within a reaction system to build a better picture of what is occurring during the reaction.
Is pressure constant in a bomb calorimeter?
A constant-pressure calorimeter measures the change in enthalpy of a reaction occurring in a liquid solution. In contrast, a bomb calorimeter 's volume is constant, so there is no pressure-volume work and the heat measured relates to the change in internal energy (ΔU=qV Δ U = q V ).
Related Question AnswersWhat is the principle of calorimetry?
A principle of calorimetry states that if there is no loss of heat in surrounding the total heat lost by hot body equal to the total heat gained by a cold body. i.e. heat loss = heat gain.What is the principle of bomb calorimeter?
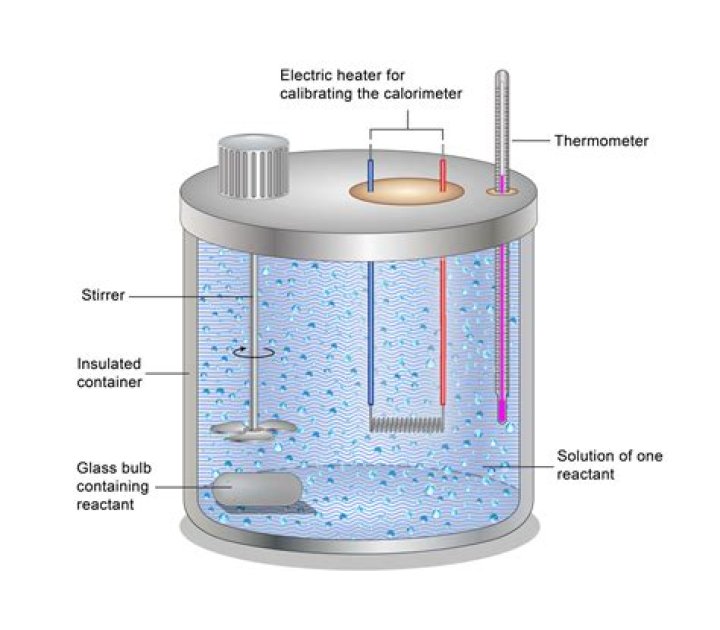
3.4 Bomb calorimetry The principle of operation is to saturate the material with oxygen, within a sealed container (the bomb) and ignite using a hot wire. During the rapid combustion carbon molecules are converted to carbon dioxide, hydrogen to water and nitrogen to gaseous nitrogen.Is work done in a bomb calorimeter?
In a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its volume is fixed and there is no volume-pressure work done. A bomb calorimeter structure consists of the following: Steel bomb which contains the reactants. Water bath in which the bomb is submerged.Why water is used in bomb calorimeter?
The water absorbs the heat, released by the combustion of the sample, resulting in a sharp rise in the water temperature (Fig. Knowing the heat capacity of the bomb calorimeter material, water, and of the fuse wire, one can calculate the exact amount of heat released by combustion of the sample.Why is bomb calorimeter more accurate?
The temperature difference of the bomb vessel is measured, just as it was for a coffee cup calorimeter. Due to the mass of the bomb vessel, time needs to be given for the reaction heat to be totally absorbed into the bomb, but still quicker than 80% of the calorimeters on the market.Who uses calorimetry?
A calorimeter is used to measure the enthalpy, the heat energy, of a reaction. Calorimeters can vary in quality. Simple calorimeters are used in high school chemistry classrooms, but much more sophisticated calorimeters are used in industry.What is water equivalent of calorimeter?
Water equivalent is generally used in comparison of a water quantity. For example, Water equivalent of a calorimeter is nothing but amount of water which will absorb same amount of heat that the calorimeter will absorb.Why is water added to a bomb calorimeter before firing?
Since we do not purge the calorimeter to remove the air in the combustion chamber, during the combustion process the nitrogen of the air forms oxides of nitrogen. It is necessary to add about 1 mL of water to the bomb to absorb the oxides of nitrogen to form HNO3.What are the two types of calorimetry?
The different calorimeters types are given below:- Adiabatic Calorimeters.
- Reaction Calorimeters.
- Bomb Calorimeters (Constant Volume Calorimeters)
- Constant Pressure Calorimeters.
- Differential Scanning Calorimeters.
What is the use of calorimetry?
A calorimeter is an instrument used in calorimetry for measuring the amount of heat released or absorbed in chemical or physical reactions. It can determine heat content, latent heat, specific heat, and other thermal properties of substances.How do you measure calorimetry?
The Calorimetry Formula- Q = heat evolved (equal to heat absorbed − heat released) in joules (J)
- m = mass in kilograms (kg)
- c = specific heat capacity in J/kg⋅°C (or J/kg⋅K)
- ∆T = temperature change in °C (or K)