Does solid have vapor pressure?
.
Besides, what is the vapor pressure of a solid?
For most solids this pressure is very low, but some notable exceptions are naphthalene, dry ice (the vapor pressure of dry ice is 5.73 MPa (831 psi, 56.5 atm) at 20 °C, which causes most sealed containers to rupture), and ice. All solid materials have a vapor pressure.
what is the difference between the pressure exerted by a vapor and the vapor pressure of a substance? The main difference between partial pressure and vapor pressure is that partial pressure is the pressure exerted by an individual gas in a mixture as if it were alone in that system, while the vapor pressure refers to the pressure exerted by the vapor in its thermodynamic equilibrium with its condensed state of liquid
Additionally, what determines vapor pressure?
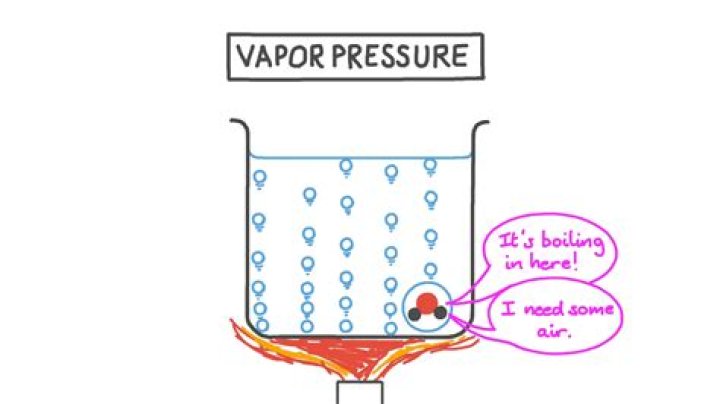
Vapor pressure is the pressure caused by the evaporation of liquids. Three common factors that influence vapor press are surface area, intermolecular forces and temperature. The vapor pressure of a molecule differs at different temperatures.
Is vapor pressure a physical property?
Vapor Pressure Evaporation is slow and thus the vapor exerts a low pressure in a closed container. Low vapor pressure is an important physical property of water, since lakes, oceans, and other large bodies of water would all tend to evaporate much more quickly otherwise. Vapor pressure is influenced by temperature.
Related Question AnswersDoes higher vapor pressure mean faster evaporation?
The greater the pressure it exerts, the weaker the intermolecular forces between molecules in its liquid state; the more volatile the liquid; the lower the boiling point and the faster its evaporation rate. The higher the vapor pressure of a material at a given temperature, the lower the boiling point.What is vapor pressure and what causes it?
When a liquid is heated, its molecules obtain sufficient kinetic energy to overcome the forces holding them in the liquid and they escape into the gaseous phase. By doing so, they generate a population of molecules in the vapor phase above the liquid that produces a pressure—the vapor pressure of the liquid.Are vapor pressure and boiling point related?
The normal boiling point is the temperature in which the vapour pressure of a liquid becomes equal to atmospheric pressure. If the intermolecular forces are small, the liquid has a high vapour pressure. More heat will be required to separate the molecules, so the boiling point will be higher.What do you mean by pressure?
Pressure is defined as the physical force exerted on an object. The force applied is perpendicular to the surface of objects per unit area. Unit of pressure is Pascals (Pa).What vapor pressure is considered volatile?
Any substance with a significant vapor pressure at temperatures of about 20 to 25 °C (68 to 77 °F) is very often referred to as being volatile. The vapor pressure of a substance is the pressure at which its gaseous (vapor) phase is in equilibrium with its liquid or solid phase.What is the unit of vapor pressure?
The most common unit for vapor pressure is the torr. 1 torr = 1 mm Hg (one millimeter of mercury). Most materials have very low vapor pressures. For example, water has a vapor pressure of approximately 20 torr at room temperature (22 °C = 72 °F).What is the formula for vapor pressure?
In chemistry, vapor pressure is the pressure that is exerted on the walls of a sealed container when a substance in it evaporates (converts to a gas). To find the vapor pressure at a given temperature, use the Clausius-Clapeyron equation: ln(P1/P2) = (ΔHvap/R)((1/T2) - (1/T1)).Does vapor pressure depend on volume?
Vapor pressure is the pressure exerted by a liquid back on the atomosphere. Vapor pressure depends on the nature of the liquid and the temperature. No matter what volume the water is, the vapor pressure of the water is the same as long as the temperature is not change.What has the highest vapor pressure?
Thus at room temperature, the substance with the lowest boiling point will have the highest vapor pressure (easiest to get into the gas phase). The substance with the highest boiling point will have the lowest vapor pressure. Vapor pressure is a liquid property related to evaporation.How does pressure affect boiling point?
Pressure Affects the Boiling Point. When atmospheric pressure increases, the boiling point becomes higher, and when atmospheric pressure decreases (as it does when elevation increases), the boiling point becomes lower. Pressure on the surface of water tends to keep the water molecules contained.What is the vapor pressure of water at 100 C?
At 100 ∘ C , the vapor pressure of water is 1 atmosphere; i.e. 100 ∘ C is the normal boiling point of water.How do you determine boiling point?
Boiling-Point Elevation Example- Glucose is our solute and water is the solvent.
- Kb for water is 0.51 degrees Celsius kg/mol.
- The molar mass of glucose is 180 g/mol.
- The boiling point of water is 100 degrees Celsius.
- The equation for boiling-point elevation is delta T = mKb.



