Does ch3cooh dissociate?
.
Hereof, does acetic acid completely dissociate?
Acetic acid is extremely soluble in water, but most of the compound dissolves into molecules, rendering it a weak electrolyte. Thus, even if a substance is not very soluble, but does dissociate completely into ions, the substance is defined as a strong electrolyte.
Secondly, can ch3cooh be hydrolysed? Being the conjugate base of a weak acid, CH3COOH, it is a relatively strong base. Thus CH3COO– accepts H+ ion from water and undergoes hydrolysis. Since Na+ is weak acid it won't undego hydrolysis.
Regarding this, what does acetic acid dissociate into?
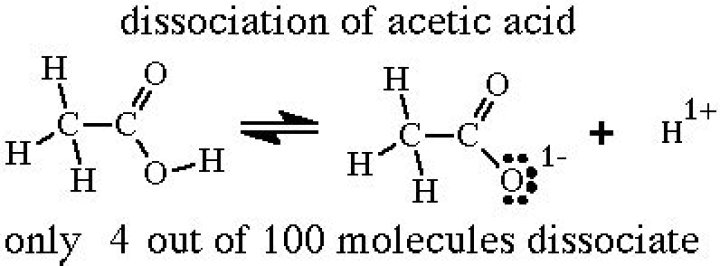
Acetic acid, formula CH3COOH, is a simple organic acid, with a pKa of 4.76. Pure water, H2O has a nominal pKa of 14 at 25*C. This means that a significant fraction of acetic acid deprotonates to the acetate ion (CH3COO-) in H2O, thereby generating an equal fraction of H3O+ (Hydronium) and CH3COO- (Acetate) ions.
Is ch3cooh a strong or weak acid?
HCl is a strong acid, and CH3COOH is a weak acid. During a reaction with a strong base, the weak acid is forced to completely dissociate (there is no weak acid molecule left intact at the equivalence point). However, the accepted value for the ∆Hrxn for CH3COOH is about 1.9 kJ/mol less than for the HCl.
Related Question AnswersDo strong acids dissociate completely?
For all practical purposes, strong acids completely dissociate in water. That is the definition: A strong acid is an acid that completely dissociates in water. For all practical purposes, HCl is completely dissociated in solution. Strong acids have a large dissociation constant, so they dissociate completely in water.Why do strong acids and bases dissociate completely?
Strong acids are in equilibrium with their conjugate bases. However, the free energy of dissociation (ie. the Ka) is so high that for all practical purposes the acid is completely dissociated. The strength of the acid is largely determined by the stability of the anion being generated.What is the world's strongest acid?
The carborane superacids may be considered the world's strongest solo acid, as fluoroantimonic acid is actually a mixture of hydrofluoric acid and antimony pentafluoride.Why do strong electrolytes dissociate?
When ionic compounds dissolve in water, the ions in the solid separate and disperse uniformly throughout the solution because water molecules surround and solvate the ions, reducing the strong electrostatic forces between them. This process represents a physical change known as dissociation.Do strong electrolytes dissociate in water?
In an aqueous solution a strong electrolyte is considered to be completely ionized, or dissociated, in water, meaning it is soluble. Strong acids and bases are usually strong electrolytes.Which is the strongest acid in aqueous solution?
H3O+ is the strongest acid that can exist in aqueous solutions. Acids stronger than H3O+ react completely with water to form H3O+ and their conjugate bases.What is dissociation reaction?
A dissociation reaction is a chemical reaction in which a compound breaks apart into two or more components. The general formula for a dissociation reaction follows the form: AB → A + B.Do acids and bases dissociate in water?
Acids and bases dissolve in water and, because they increase the concentration of one of the products of water self-ionization, either protons or hydroxide ions, they suppress water dissociation. For any acid, Ka is the equilibrium constant for the acid dissociation reaction in water.What is the dissociation reaction for vinegar?
The equation for the dissociation of acetic acid, for example, is CH3CO2H + H2O ⇄ CH3CO2− + H3O+.How do you find the dissociation constant?
1 Answer- Write the equation for the dissociation of a generic monoprotic acid: HA + H2O ⇌ H3O? + A?
- Write the dissociation constant expression. Ka=[H3O?][A?][HA]
- Determine the equilibrium concentrations. pH = -log[H3O?] = 4.69.
- Substitute these values in the expression and that is it!