Can two metals form a compound?
.
Similarly one may ask, can two metals form a molecular compound?
Pairs of atoms of some elements also form covalent bonds between themselves. These are molecules, because they are two atoms held together with a covalent bond, but they are still elements, because the atoms are of the same type, and the molecule retains the properties of the element.
| Prefix | Number |
|---|---|
| 10 | deca |
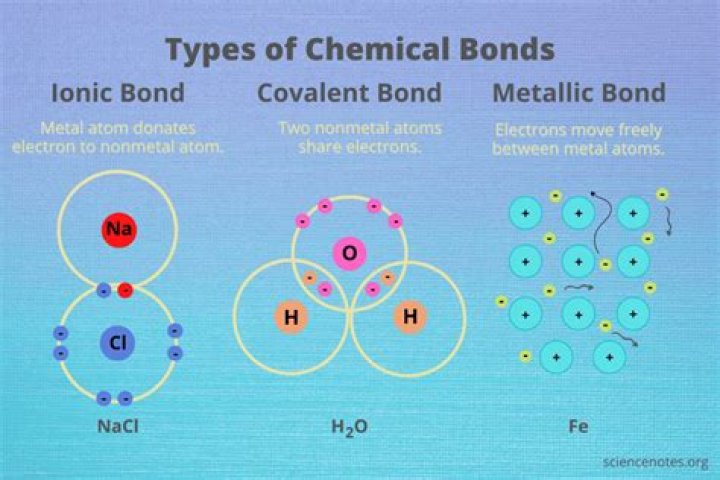
Likewise, what kind of compound is two metals? A binary compound is formed from two types of elements - their states of matter result in the following types of compound : Metal + Metal = Metallic Compound. Metal + Non-Metal = Ionic compound.
Also, why can't two metals form a compound?
Explanation: Although there definitely is such a thing as metallic bonding, when we combine two or more metals, the result is a mixture. This means that there is no definite proportion to the combination, the way there is for a compound. This cannot be done with a compound.
Can two metals form a covalent bond?
Ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when electrons are shared between two nonmetals. A covalent bond involves a pair of electrons being shared between atoms. Atoms form covalent bonds in order to reach a more stable state.
Related Question AnswersWhat is it called when two metals are mixed together?
Metallic bonds can occur between different elements. A mixture of two or more metals is called an alloy. Depending on the size of the atoms being mixed, there are two different kinds of alloys that can form: Substitutional alloy.What makes a compound metallic?
A metallic compound is a compound that contains one or more metal elements bonded to another element. Typically, the metal atom acts as the cation in the compound and is bonded to a nonmetallic anion or an ionic group. When metals bond to other metals, they form an alloy.How do you form a compound?
Compounds form when atoms chemically combine. When metals combine (react) with nonmetals, ionic compounds usually form. When nonmetals combine, covalent compounds usually form. Molecules are the smallest part of a compound that have the properties of the compound.What is a molecular compound?
A molecular compound can be defined as a compound where the atoms share electrons through covalent bonds. It is also known as a covalent compound. The covalent bonds are what hold the molecule together. When atoms share electrons, they are able to have a full outer electron shell.How do metals combine?
Forming ionic compounds. Metal atoms have only a few electrons in their outer shell whereas non-metal atoms have lots of electrons in their outer shell. This means that metals tend to react with non-metals. When a metal reacts with a non-metal, electrons transfer from the metal to the non-metal.Is hydrogen a metal?
Hydrogen is not considered a metal, even though it does exist in Group I (alkali metals) of the periodic table.Why do metals bond?
In metallic bonds, the valence electrons from the s and p orbitals of the interacting metal atoms delocalize. That is to say, instead of orbiting their respective metal atoms, they form a “sea” of electrons that surrounds the positively charged atomic nuclei of the interacting metal ions. Metals are shiny.What a compound is?
A compound is a substance formed when two or more chemical elements are chemically bonded together. Example 1: Pure water is a compound made from two elements - hydrogen and oxygen. The ratio of hydrogen to oxygen in water is always 2:1. Each molecule of water contains two hydrogen atoms bonded to a single oxygen atom.What is the smallest part of a compound?
The smallest particle of a compound capable of existing on its own is a Molecule-not a atom. An atom is the smallest particle capable of existing on its own of an ELEMENT. And yes,atoms can be further broken down into electrons,neutrons and protons.How do you combine elements?
Elements primarily combine to form compounds through two main types of chemical bonding: ionic bonding and covalent bonding. A compound is any molecule that is made up of two or more different elemental atoms. In order for most atoms to become stable, they must bond with other atoms that are available.Why do compounds form?
Compounds form from the elements' exchanging of electrons. For instance, sodium has an extra electron it wants to get rid of. While chlorine badly needs an electron to fill its outer shell. When these two elements combine, the electron is traded over and both elements are satisfied.What is another name for a covalent compound?
A covalent bond, also called a molecular bond, is a chemical bond that involves the sharing of electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.How do atoms combine?
Most interactions among atoms take place in the outermost shell of each atom. The number of each electron in this shell determines how an atom combines with other atoms to form compounds. When atoms combine they gain, lose or share electrons in such a way that the outer shells become chemically complete.Is gold a pure substance?
They are: Pure Substance: The substances that are free from any kind of mixture and contain only one kind of particle are pure substances. Examples of pure substances include iron, aluminum, silver, and gold.What are 2 types of compounds?
There actually are simple These two types are called "molecular" compounds and "salts" (or equivalently "ionic" compounds): Molecular compounds: These compounds are made up of molecules whose atoms bind to one another through "covalent" bonds. What are some examples of elements and compounds?What is a Type 3 compound?
Type III binary compounds contain no metal atoms. There are two different naming systems for Type III binary compounds: the “old system” and the “new system.” The old system uses prefixes to indicate the number of each atom present and the new system is identical to that used for naming Type II compounds.What are the 4 types of compounds?
There are four types of compounds, depending on how the constituent atoms are held together:- molecules held together by covalent bonds.
- ionic compounds held together by ionic bonds.
- intermetallic compounds held together by metallic bonds.
- certain complexes held together by coordinate covalent bonds.



